Fujifilm Unveils New DR Technology to Orthopedists
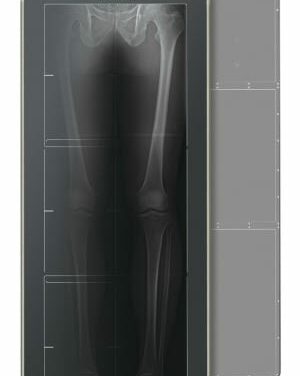
Fujifilm Medical Systems will showcase its FDR D-Evo GL DR detector for single-exposure, long-length digital imaging at this week’s American Association of Orthopaedic Surgeons Annual Meeting in Orlando. The world’s first long-length digital radiography (DR) detector, the D-Evo GL has recently received FDA 510(k) clearance and will be readily available in the United States this spring.
Read More