|
Sonora Medical Systems Inc
Sonora Medical Systems Inc, Longmont, Colo, objectively tests and ensures the proper operation of medical imaging devices that deliver accurate morphologic and physiologic information. From this information, physicians make diagnosis and treatment decisions. These devices have become more technologically complex and able to detect disease at the earliest stages. The company relies on a process called evidence based performance assurance (EBPA), and tools such as FirstAssist. EBPA gives patients, sonographers, and physicians confidence that imaging systems are performing at optimal levels and producing quality diagnostic information.
(888) 476-6672 ext 681
www.4sonora.com
Ambassador Medical
 |
Ambassador Medical, Carmel, Ind, is a GE Healthcare company that provides high-quality preowned ultrasound equipment. Its inventory includes not only systems but also a wide variety of transducers and parts from almost every make and model. Services include repairs to circuit boards, power supplies, back end processors, and peripherals. Recently, Ambassador was recommended to receive ISO 13485 for medical devices. This accomplishment solidifies its commitment to quality.
(888) 499-4554
www.ambassadormedical.com
Conquest Imaging
 |
The 24/7 technical support staff at Conquest Imaging, Stockton, Calif, reports that on some occasions, GE back end processor (BEP) equipped systems will give a warning message of “No Archive.” The problem is that the TCP/IP looks correct, but has been corrupted and will not work.
The solution is to change the last digit in the IP address by 1. For example, if it is .234, change it to .235, save the setting, and reboot. Then change it back to the original setting, and then save the setting and reboot. This is a known bug in BEP-equipped systems and especially prevalent in the Vivid 7.
(800) 496-9906
www.conquestimaging.com
 |
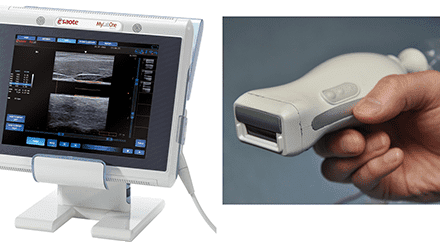
Siemens Medical Solutions
The Acuson P10, a pocket ultrasound system from Siemens Medical Solutions, Malvern, Pa, is a handheld diagnostic device that is a little larger than a typical PDA, weighs 1.6 pounds, and fits in a lab coat pocket. The device is intended for complementary initial diagnostic care and triage, particularly in cardiology, emergency care, and obstetrics, and is useful for physicians and medical personnel in intensive care units, ambulances, and helicopter air medical transports. It helps detect conditions that may be clinically significant but have previously required expansive or invasive diagnostic testing in asymptomatic patients. The device has received FDA 510(k) clearance.
(888) 826-9702
www.pocketultrasound.com