By Brian Bean, CRES
 Brian Bean, CRESSingle photon emission computed tomography (SPECT) has been around longer than newer technologies like MRI and PET, but it does not share their name recognition. Given radioisotope shortages in recent years, some wonder if it even has a future—but do not count it out yet. There are advances on the horizon promising to bring SPECT back into the spotlight, ensuring its position as a valued member of the molecular imaging family.
Brian Bean, CRESSingle photon emission computed tomography (SPECT) has been around longer than newer technologies like MRI and PET, but it does not share their name recognition. Given radioisotope shortages in recent years, some wonder if it even has a future—but do not count it out yet. There are advances on the horizon promising to bring SPECT back into the spotlight, ensuring its position as a valued member of the molecular imaging family.
This article will provide a brief overview of SPECT and will look at some of the latest developments, including advances in domestic radioisotope production. Understanding SPECT will help clinical engineering professionals of all backgrounds stay informed and enable them to stay on top of the rapid changes taking place in the health care industry today. It may also further your interest in the field and offer an expanded career path.
Before introducing SPECT, let us first look at planar nuclear medicine (NM) because SPECT is to NM what computed tomography (CT) is to x-ray. An x-ray image is produced when x-rays pass through a patient onto an imaging receptor such as film or a digital detector. X-ray photons are attenuated by dense bones more so than other tissues, resulting in a 2D image of the anatomy with contrast levels varying in proportion to the number of x-rays received. CT improves on this by obtaining x-rays that are a full 360o around a patient as a series of thin 2D images or “slices” of anatomy that can be viewed individually or reconstructed into a 3D image.1
In planar NM, a 2D image is produced by gamma rays picked up by the detector of a “gamma” camera, which are emitted from a patient who has been given a radiopharmaceutical. Gamma rays are similar to x-rays, but are produced in the nucleus of an unstable “radionuclide” atom as it decays. So while NM can visualize bones in the body, it does so through a different process than x-ray. In the case of a bone exam, NM visualizes increased areas of bone turnover, which can indicate arthritis, swelling, or even a malignant tumor.
Expanding Nuclear Imaging
SPECT improves on planar NM by rotating the detector around the patient while acquiring a series of tomographic projections. This greatly improves the image contrast resolution and allows for further 3D reconstructing. Typical SPECT exams include a brain scan for someone suffering from severe headaches or to check for a stroke; a cardiac exam to measure the output of the heart, visualize arteries, and show blood flow within the heart muscle itself; and exams of organs such as the lungs, kidneys, liver, and gall bladder to check for proper blood supply or abnormalities such as cysts or tumors. This, however, is only the beginning. In addition to visualizing the overall condition of an organ, specific physiological and metabolic processes can also be imaged down to the molecular and cellular levels.
Cardiac exams are typically performed with the two detectors in the 90-degree configuration shown so that two images can be simultaneously acquired as the detectors rotate around the patient, reducing scan times by half.
SPECT excels at showing physiology but lacks detailed anatomical information, which can make it difficult to pinpoint exactly where areas of activity are occurring within a patient. Historically, a SPECT image would be used together with a CT image to localize areas of interest. Trying to compare two different images from two different machines can be difficult, expensive, and time-consuming. A solution to this problem is today’s new dual modality SPECT/CT machines.
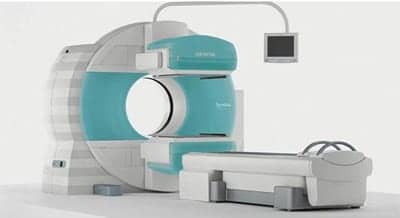 Siemens Symbia SPECT cameras are available with or without the CT option. The dark ring that is visible here on the inside of the gantry is the x-ray port for the CT, indicating this is a SPECT/ CT combination gantry.
Siemens Symbia SPECT cameras are available with or without the CT option. The dark ring that is visible here on the inside of the gantry is the x-ray port for the CT, indicating this is a SPECT/ CT combination gantry.
These machines allow the acquisition of a low-dose CT—because it does not have to be diagnostic quality—to be used to apply anatomical information to the physiological information from a SPECT exam.2 Combining the physiological information from SPECT in color with the grayscale anatomical information from CT produces an image where areas of metabolic activity immediately stand out. The high-quality diagnostic images produced by the relatively low-cost combination machines are making these units very popular.
Here a dual SPECT/CT camera illustrates how the physiological SPECT image (left) and the anatomical CT image (middle) can be combined to form a high-quality diagnostic image (right). Shown here by the arrows is a nodule of interest located just off the tip of the spleen.
Learning the Equipment
Upon entering a nuclear exam room, you will see the two main components of a gamma camera: A gantry and the patient table.3 The gantry typically has two or three detectors that rotate 360o around the patient and move radially in and out. Newer detectors have infrared look-ahead sensors that dynamically drive the detectors in to follow the contours of the patient to keep them as close as possible for the best-quality images. Patient tables do not have a lot of movements; they move up and down for easy patient access and the tabletop indexes fully through the gantry, allowing for complete head-to-foot coverage of the patient.
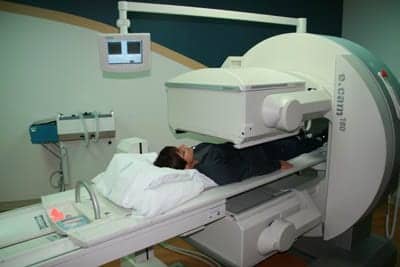 This dual-head Ecam is set up for a whole-body bone scan with the two detectors facing each other. Note the Xenon unit in the background. This is used for dispensing Xenon-133 gas for lung exams.
This dual-head Ecam is set up for a whole-body bone scan with the two detectors facing each other. Note the Xenon unit in the background. This is used for dispensing Xenon-133 gas for lung exams.
On the face of a detector is a removable collimator, and different collimators are used for different energy isotopes and resolutions. With the collimator turned off, there is an aluminum sheet that protects a large, delicate thallium doped sodium iodide crystal. This crystal scintillates or produces light when excited by a gamma ray; the light is picked up and amplified by photomultiplier tubes (PMTs) that are optically coupled to the crystal and an 18 x 24-inch detector can have as many as 50 3-inch-diameter PMTs to cover the entire crystal. A PMT is a relatively large vacuum tube that detects the weak light emitted from the crystal and amplifies it into a usable voltage signal. The electrical signals are then digitized and sent to a workstation for further processing. Much of the calibrations and diagnostic procedures on cameras today are automated and under software control.
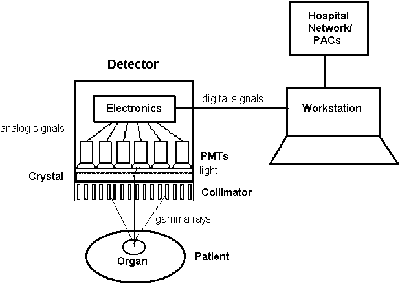 A simplified overview of a nuclear camera system.3 The primary reason that nuclear detectors are so large is because of the size and number of vacuum tube PMTs required. (Click to enlarge.)
A simplified overview of a nuclear camera system.3 The primary reason that nuclear detectors are so large is because of the size and number of vacuum tube PMTs required. (Click to enlarge.)
Vacuum tube PMTs have been around for years, but they suffer from being large, delicate, and sensitive to magnetic fields such as in an MRI. Possible replacements are being explored, such as Silicon Photomultipliers (SiPMs). These are basically a matrix of reverse biased photosensitive diodes that operate in the “avalanche” mode. When a light photon strikes a diode it ejects an electron, starting a small current flow that quickly cascades or avalanches, converting a single light photon into a usable voltage signal. Some problems like high costs and slow recovery times must be overcome before SiPMs can become widely accepted.4
Isotopes
Once a CT machine is plugged in, it can produce x-rays and is essentially ready to go. A SPECT scanner, on the other hand, requires a constant supply of radionuclides. Recent shortages have focused a lot of attention on the fact that there is currently no large-scale production of medical isotopes in the US. The entire world supply is produced by the few World War II-era nuclear reactors still in operation. These reactors are now experiencing increasing downtimes due to maintenance and repairs to the extent that the US has experienced delays, postponements, and even cancellations of patient exams. However, breakthrough technologies are promising to overcome the long-standing problems associated with producing radionuclides without a nuclear reactor.5
The American Medical Isotopes Production Act of 2011, a bill to promote the production of molybdenum-99 (Mo-99) in the US, was passed unanimously by the senate in December 2012,6 ensuring there will be continued attention and funds available for the research and development into new technologies being done by several startup companies in the US. These technologies include a neutron beam driven fusion/fission technology, a patented chemical separation process, and others, all of which have closely guarded proprietary methods.7,8
It should be noted that the Mo-99 mentioned in the bill is “parent” isotope of Tc99m. When Mo-99 decays, it becomes Technetium 99 metastable (Tc99m). The benefit of this is Mo-99 has a 66-hour half-life—long enough for it to be transported. The 6-hour half-life of Tc99m is too short for transport, but allows enough time for imaging patients, while at the same time limiting radiation exposure to patients. As a side note, it should be mentioned that positron emission tomography (PET) also uses radioactive materials for imaging. However, PET isotopes are of higher energy and have shorter half-lives—some on the order of minutes—and therefore need to be obtained locally. This is not a huge problem because they can be produced with a cyclotron and there are many production and distribution centers located across the US.
Hot Labs
Tc99m is the radionuclide used in a large majority of the 13 million nuclear exams done annually in the US. Tc99m mixes readily with a wide assortment of pharmaceuticals to produce the end product radiopharmaceutical that is administered to the patient. Dozens of Tc99m-based radiopharmaceuticals exist today that are formulated to target specific biological functions, with new ones being developed all the time. The storage and handling of Tc99m and other radioactive materials is done in dedicated rooms called Hot Labs. These areas usually do not get a lot of attention, but increasing regulations are bringing them into the spotlight.
Hot Labs are now being required to adhere to USP Pharmacopeia chapter 797, pharmaceutical compounding—sterile preparations, part of which includes complying with the International Organization for Standards (ISO) guidelines that include requiring a positive pressure air supply in areas where materials are compounded.9 In some situations, this can be achieved by using a tabletop air handler. Once a patient dose has been made up and administered to the patient, typically through injection although it can be ingested or inhaled, and has had time to distribute within the body, the patient is ready to be scanned.
 This tabletop air handler blows filtered air down into the work area, creating an ISO level 5–compliant environment for working with sterile compounding materials. Also shown is a dosimeter for measuring dose strengths. Samples are lowered into the round column for measurement by a single PMT.
This tabletop air handler blows filtered air down into the work area, creating an ISO level 5–compliant environment for working with sterile compounding materials. Also shown is a dosimeter for measuring dose strengths. Samples are lowered into the round column for measurement by a single PMT.
Future Advances
Hopefully, this brief overview has helped to introduce the lesser known, continually evolving SPECT modality and shown some of what we can expect to see in the near future in medical radioisotope production here in the US. Not only do domestically produced isotopes reduce the proliferation of nuclear materials, they also help ensure that SPECT research and development can continue without the fear of being shut down due to a lack of radioisotopes.
Some advancements we are likely to see in the near future include a SPECT/MRI machine, once there is a suitable replacement for magnetically incompatible PMTs; new improvements in collimators that promise to increase spatial and contrast resolutions; and new software algorithms that can reduce processing times while improving image quality at the same time.
On the clinical side, we can expect to see advances in nuclear pharmacology where it is possible to track the uptake of new drugs to test their efficacy; nuclear psychiatry, or the mapping of neurotransmitter activity in the brain in relation to behavior and disease; the tagging of stem cells; new gene therapies; and more.10 We have only begun to scratch the surface of the possibilities ahead. 24×7 Service Solutions May 2013
Brian Bean, CRES, clinical engineering, Baystate Medical Center, Springfield, Mass, has more than 20 years of experience in the biomedical field and more than 10 years in nuclear medicine and diagnostic imaging. For more information, contact [email protected].
References
1. Rahmim A, Zaidi H. PET Versus SPECT: Strengths, Limitations and Challenges. Lippincott Williams and Wilkins; October 2007.
2. Miller JC, Phil D. Hybrid SPECT/CT: Enhancing nuclear medicine. Radiology Rounds. Manchester General Hospital. 2010;8(7).
3. Bean B. Demystifying nuclear medicine. Imaging Horizons. Association for the Advancement of Medical Instrumentation, 2009.
4. Tao A. Development of a silicon photomultiplier-based gamma camera. McMaster University; Hamilton, Ohio; October 2011.
5. Future alternatives to Molybdenum-99 production for medical imaging. Canadian Agency for Drugs and Technologies in Health; January 2011.
6. Le T. Congress passes American Medical Isotope Production Act. Imaging Biz News. December 27, 2012.
7. Northstar’s Technology. Northstar Medical Radioisotopes LLC. Available at: http://www.northstarnm.com. Accessed April 9, 2013.
8. Shine Medical Technologies. Available at: http://shinemed.com. Accessed April 9, 2013.
9. Welcome to USP797.org. usp797.org inc. Available at: http://www.usp797.org. Accessed April 9, 2013. 10. Advancing nuclear medicine through innovation. Committee on State of the Science of Nuclear Medicine; National Academy of Sciences; Washington DC, 2007.




