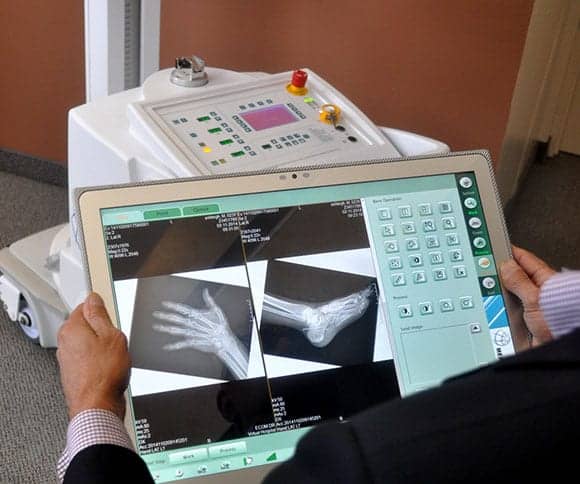
Meridian Medical Systems, Aurora, Ohio, recently announced that its Monarch ultra high definition (UHD) universal digital imager (UDI) upgrade with wireless detector and automatic exposure detection (AED) has been cleared by FDA for marketing in the United States. According to the company, the software is specified to operate as a certified component when used with the Meridian operator’s console to control the x-ray generator and other acquisition and display parameters.
When used with a 20-inch (4K) acquisition console featuring 3840 x 2560 display resolution (9.8 mp), the Monarch UDI 2 with UHD creates images at a resolution typically seen only on high-resolution diagnostic workstations, according to the company. Meridian says its UDI can also display images twice as fast as most other workstations.
“The FDA granted clearance based on a full range of high-performance qualities, including system specifications, quality system management, dose parameters for pediatrics, and non-clinical test data and performance for solid-state detectors,” said Larry Cornell, president of Meridian Medical Systems.
According to Meridian’s website, the company’s engineers have integrated most manufacturers’ generators into the core communication protocols for the UDI to improve workflow and throughput capabilities. The techniques and acquisition protocols are automatically transferred from the RIS, to the Meridian Acquisition Workstation, to the x-ray room’s generator. Meridian reports that its UDI Smart Digital Upgrades can be configured with any combination of up to 3 flat panel detectors. All panels supported by Meridian are said to offer superior image quality and lowest dose, and can be fixed, tethered, or wireless.
For more information about this and other Meridian UDI x-ray room digital upgrades, visit the Meridian Medical Systems website.