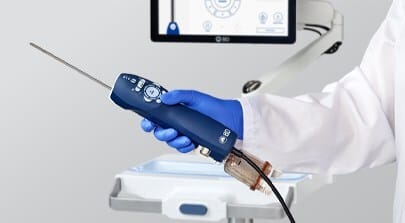
Fresenius Medical Care, a provider of technologies for individuals with renal diseases, has obtained FDA 510(k) clearance for its VersiHD with GuideMe Software. The advanced self-guided interface, integrated into the VersiHD chronic home hemodialysis system (HHD), is poised to reshape the experience of home hemodialysis for both patients and healthcare professionals.
VersiHD with GuideMe Software provides graphical walk-through guidance that aims to enhance ease of use and confidence for patients and nurses. It is designed to improve patient training time, ease the transition to home, and make the training experience easier for new users.
“We expect VersiHD with GuideMe Software to further simplify home hemodialysis for patients during training and at home,” says Brigitte Schiller, MD, senior vice president, medical officer, Home Therapies at Fresenius Medical Care. “As a result of this software upgrade, patients and their care partners will have additional support to be confident with the therapy at home. VersiHD with GuideMe Software will support Fresenius Medical Care’s mission of advancing access to home therapies to more patients.”
VersiHD with GuideMe Software will initially be available in selected markets in 2023. Existing VersiHD systems will be upgradeable to GuideMe Software. For more information about VersiHD with GuideMe Software, please visit www.nxstage.com/hcp/versihdguideme
NxStage from Fresenius Medical Care, draws on its experience of over 30 million patient treatments at home over nearly two decades, serving patients in more than 45,000 homes with a broad diversity of water sources across the U.S.
NxStage has had many significant innovations over the years guided by patient input starting with the first portable HHD machine, NxStage System One, in 2005 followed by the introduction of the PureFlow DI Water Purification System in 2006. Nx2me Connected Health, the first Connected Health platform for HHD followed in 2014.
The NxStage System One was the first machine to receive FDA clearance for Nocturnal HHD indication in 2014 and also the first to receive FDA clearance for Solo HHD indication in 2017. In 2017, NxStage introduced the VersiHD touchscreen cycler. Over 95% of HHD patients in the U.S. use the NxStage HHD system, according to Fresenius.