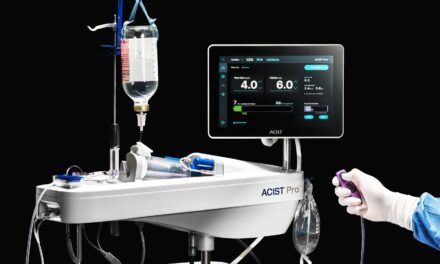
The platform combines ultrasound and optical imaging to provide detailed views of coronary anatomy in a single workflow.
Conavi Medical has received Food and Drug Administration (FDA) 510(k) clearance for its next-generation hybrid imaging system. The platform is designed to provide a detailed assessment of coronary anatomy within a single workflow by integrating two distinct imaging technologies.
The system combines intravascular ultrasound (IVUS) and optical coherence tomography (OCT) into a single platform. These technologies enable physicians to visualize deep vessel structures and high-resolution surface detail in real time, according to a company release. The technology is intended to support physician decision-making and streamline clinical workflows.
“FDA clearance of our next-generation hybrid imaging system marks a pivotal moment for Conavi as we transition into commercial launch in the US,” says Thomas Looby, chief executive officer of Conavi Medical, in a release. “We believe hybrid IVUS-OCT imaging represents the next evolution in intravascular imaging, bringing together IVUS and OCT into a single system to give a complete picture of the vessel.”
Advancing Intravascular Imaging
The system allows for simultaneous, co-registered IVUS and OCT imaging in a single pullback. It also includes analysis tools for lesions and stents that incorporate insights from both imaging modalities, according to the company. The design features a catheter intended for enhanced deliverability and a streamlined connection workflow.
“Hybrid IVUS-OCT imaging has the potential to meaningfully enhance how we approach complex coronary interventions,” says Megha Prasad, MD, interventional cardiologist at New York-Presbyterian Hospital, in a release. Prasad notes that the ability to evaluate plaque composition, vessel size, and stent expansion in a single pullback can support procedural decision-making and efficiency.
Intravascular imaging is increasingly used in percutaneous coronary intervention, as clinical evidence shows improved outcomes when imaging guides stent sizing and placement. However, the company states that global adoption remains limited due to workflow complexity and the historical need for multiple imaging systems.
Market Growth and Commercial Launch
The global market for coronary intravascular imaging is estimated to exceed $4 billion annually, with current penetration at 30% to 40% of eligible procedures, according to Conavi Medical. Growth in this segment is driven by an increasing focus on optimizing stent placement and the integration of advanced imaging into clinical workflows.
Following the regulatory clearance, the company is preparing for commercialization in the US. Conavi Medical expects to initiate a limited market release in select US centers during the second half of 2026.
Photo caption: The next-generation hybrid imaging system combines intravascular ultrasound (IVUS) and optical coherence tomography (OCT) into a single platform.
Photo credit: Conavi Medical