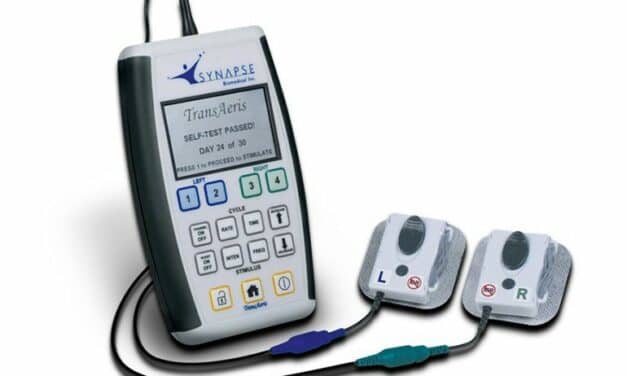
FDA Grants Breakthrough Designation to Synapse Biomedical’s TransAeris System
Ohio-based Synapse Biomedical, Inc. announces that the U.S. FDA has granted Breakthrough Therapy Device designation to TransAeris, a temporary percutaneous intramuscular diaphragm stimulator designed to aid in weaning from mechanical ventilation.
Read More