
Tobey Clark, CCE
It has been 5 years since The Joint Commission Clinical Alarm National Patient Safety Goal was removed as a hospital requirement. Are clinical alarms less of an adverse event issue now than they were in 2005, or are events increasing? Are the problems similar or of a different nature? Have new facility designs impacted alarm improvement? What about noise reduction? What effect has technology had on alarm safety? Are bedside alarms more reliable? Have wearable alarm annunciators—such as phones and pagers connected through monitoring networks, specialized alarm integration engines, and the telecommunications system—been beneficial?
Well, if you have been reading the newspapers, you will likely say the situation is worse. On February 21, 2010, The Boston Globe published a front page article “MGH Death Spurs Review of Patient Monitors.”1 The lay press story stated, “device issues spotlight a growing national problem.” The Massachusetts General Hospital biomedical engineering team responded quickly and professionally to disable the alarm off setting on 1,100-plus monitors, installing distributed alarm speakers, and other measures. A July 31, 2010, article in The New York Times headlined, “For No Signs of Trouble, Kill the Alarm.”2 The article discussed numerous fatal events in other industries where alarms were turned off because they were annoying or did not really signal a true emergency hazard. A lesson should be taken from the shepherd boy in Aesop’s Fables who falsely cried “wolf” so often that when a real wolf appeared, no one responded to his “alarm.”
James Keller, VP at ECRI Institute, was interviewed for the New York Times story and aptly commented that alarms had to be smarter, crying wolf less often. He cited data from the recent ECRI Institute Webinar on alarm fatigue where two-thirds of the attendees polled stated that they had been desensitized to important clinical alarms.
Studies have shown that a large percentage of alarms do not result in a change in patient care. Nuisance alarms continue to be the most common factor leading to alarm fatigue and missed true alarms. In a 2010 paper,3 about 40% of all alarms did not correctly describe the patient condition and were classified as technically false; 68% of those were caused by patient manipulation. Only 15% of all alarms were considered clinically relevant.
Do these publicized events and articles represent the cumulative data, or are they anecdotal? At the last FDA/Medsun Webinar in 2007 on alarms,4 Kathy Weil, RN, MS, reported alarm-related deaths in the FDA Manufacturer and User Facility Device Experience—or MAUDE—database increased from 81 in 2005 to 109 in 2006. In a follow-up April 2010 ECRI Institute Health Devices safety note related to the Boston Globe article, it reported 10% of the problem summaries submitted to the institute involve alarms. Clinical alarms have been first or second as a Top Ten Health Technology Hazard over the past 3 years in ECRI Institute’s annual list. FDA and ECRI Institute both put physiological monitoring systems at the top of the causative device list, with ventilators and infusion devices also common.
The Healthcare Technology Foundation’s (HTF) 2006 white paper, “Impact of Clinical Alarms on Patient Safety,”5 provided key background, problem reporting, and improvement information along with important observations and recommendations. A national survey, which was part of the study, clearly showed that false positive, or nuisance, alarms were the biggest issue. In addition to the report’s location on the Web, hard copies were distributed at national nursing and engineering meetings and to all American College of Clinical Engineering (ACCE) members. A 2010 survey by ACCE of its membership found that slightly more than half (52%) of those completing the survey had read the HTF report. Responders reported that relations with nursing (30%) and IT (22%) were enhanced through this action. Other positive aspects included implementing alarms training (30% to 50%) and an increased emphasis on the evaluation of alarms in the purchase of new equipment (37%). Related to networking, one-third have implemented remote personal alarms, such as pagers, and increased their interaction with IT regarding interfaces and communications (39%).
Ideally, alarms would detect all impending and occurring life-threatening situations while providing diagnostic information and discrimination between patient and equipment alarms. The “front end” is of key importance in reducing nuisance alarms and other alarm “artifacts.” A quality signal must be complemented by appropriate signal processing, and by the recognition of artifacts, including movement, electromagnetic interference, and patient care procedures—the last item being critical in the reduction of nuisance alarms.
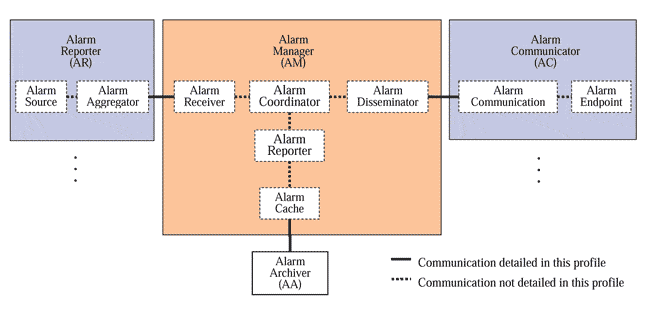
The IHE-PCD Alarm Communication Management profile provides interoperable messaging from medical devices to clinicians and appropriate responses along with the ability to archive this data.
Image courtesy of Integrating the Healthcare Enterprise (IHE)
Traditional threshold alarms are sensitive but not specific and produce an abundance of false positives. In general, “smart alarm” technological progress has not kept pace with overall medical device advances, in part due to manufacturer reluctance—liability concerns and business factors are at the top of the list. An important aspect in the development of “smart alarms” is the use of multiple parameter monitoring, common in the OR and ICU settings, to evaluate the validity of a single parameter alarm. A second area for bedside monitoring where progress is needed to reduce alarm issues is human factors design—controls, alarm setting, displays, and sounds. Standards for alarm systems exist, such as IEC 60101-1-8, but are not commonly utilized in the United States. The IEC standards specify alarm sounds based on scientific data.
Beyond the patient monitor comes the network. Traditional ICU monitoring systems have a central station. Today, the network alarms’ travel may include database servers, LANs, enterprise networks, alarm integration engines, and various output devices including telecommunication systems and remote annunciators. Integrating the Healthcare Enterprise, or IHE, has developed a patient care device technical framework draft on “Alarms Communication Management.”6 Alarm integration systems taking data from devices and device networks and distributing alarms via IP cell phones and other telecommunications devices are true examples of the convergence of health care technology. Some of the systems have programmable integrated alarm decision-making, validation, prioritization, and data storage for retrospective analysis. In the past, the FDA has stated that alarm pagers and phones could not be used as a primary means of alarm notification. Recently, the FDA has proposed to classify alarm integration systems as medical device data systems (MDDS), Class II devices requiring a 510(k). However, few vendors, including market leaders, have achieved 510(k) acceptance. Mobile alarm notification devices are most effective if key, prioritized data is displayed to the clinical caregiver and two-way communications are available.
A systematic approach such as what Johns Hopkins Hospital took in a recent article in the January 2010 edition of the American Journal of Critical Care, “Monitor Alarm Fatigue: Standardizing Use of Physiological Monitors and Decreasing Nuisance Alarms,”7 greatly reduced nuisance alarms, resulting in a 43% reduction in critical monitor alarms. In general, alarm improvement measures that show some success in enhancing clinical alarm response and actions are:
- Change alarm default settings to more clinically actionable settings—eg, expand alarm limit settings, delay alarm activation, and reduce low-priority arrhythmia settings;
- Add secondary alarm systems such as alarm pagers, strategically placed alarm annunciators, and enhanced audio and visual systems;
- Employ unit-specific and institutional care standards for alarms—responsibility and accountability—with a clear delineation of actions to be taken; standardization on a specific monitoring system; and
- Training of staff in best practices for parameter acquisition—eg, ECG technique, understanding alarm problems including fatigue, alarm setup, response to critical alarms, and protocols.
There have been some rumblings that The Joint Commission will reinstate clinical alarm safety as a hospital National Patient Safety Goal. Whether this happens or not, the positive impact of technology and clinical alarm policies has not overcome the complexity related to clinical alarm adverse events.
Don’t cry wolf! Lead the charge at your institution to find your clinical alarm gaps. Review your system, take carefully measured actions to make alarm settings at clinically actionable levels, use technology to enhance notification and clinical action, train your staff, set institutional-wide policy, and measure your results so improvements can be made.
Tobey Clark, CCE, is the director of instrumentation and technical services and adjunct faculty, engineering and nursing/health sciences, at the University of Vermont, Burlington. For more information, contact .
References
- MGH death spurs review of patient monitors. Available at: www.boston.com/news/health/articles/2010/02/21/mgh_death_spurs_review_of_patient_monitors. Accessed August 17, 2010.
- For no signs of trouble, kill the alarm. Available at: www.nytimes.com/2010/08/01/weekinreview/01wald.html?_r=1&ref=todayspaper. Accessed August 17, 2010.
- Siebig S, Kuhls S, Imhoff M, Gather U, Schölmerich J, Wrede CE. Intensive care alarms—how many do we need? Critical Care Medicine. 2010;38(2):451-456.
- Medsun clinical engineering Webcast: Clinical alarm systems and their management. Available at: medsunaudioconference.s-3.net/alarms_Nov07/index.aspx. Accessed August 17, 2010.
- ACCE Healthcare Technology Foundation whitepaper: Impact of clinical alarms on patient safety. Available at: thehtf.org/White%20Paper.pdf. Accessed August 17, 2010.
- IHE patient care device (PCD) technical framework supplement 2008-2009: Alarm Communication Management. Available at: www.ihe.net/Technical_Framework/upload/~.pdf. Accessed August 17, 2010.
- Graham KC, Cvach M. Monitor alarm fatigue: Standardizing use of physiological monitors and decreasing nuisance alarms. Available at: ajcc.aacnjournals.org/cgi/content/abstract/19/1/28. Accessed August 17, 2010.





