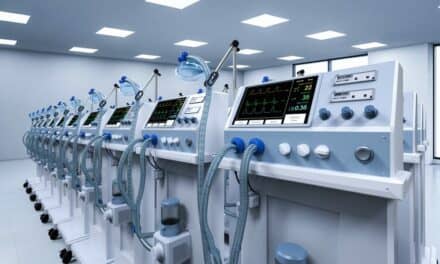
CorVent Medical, New York, N.Y., announced that the FDA has issued an emergency use authorization (EUA) for primary critical care use of the company’s RESPOND-19 Ventilator. A new type of ventilator, RESPOND-19 is designed for rapid expansion of critical care ventilation capacity to allow hospitals to treat more patients suffering from acute respiratory distress syndrome (ARDS) during times of greatest need.
The surge support system is optimized for long-term storage and ensures cost-efficient healthcare preparedness. With EUA granted, the RESPOND-19 Ventilator is now available for commercial use in the United States and the company is expecting CE Mark in early 2021.
“Like many fellow healthcare providers, I experienced the first wave of COVID-19 cases and the resulting surge in ventilator support required for patients. When demand exceeds ventilator supply, healthcare providers need the ability to quickly scale resources in a safe and financially responsible manner,” says Patrick Troy, MD, pulmonologist at Hartford Hospital in Hartford, Conn., and chief medical officer of CorVent Medical. “Moving forward, successful healthcare disaster preparation will inevitably include reliable surge support ventilators that can be deployed on-demand to meet an increased patient demand.”
The CorVent RESPOND-19 Ventilator complements existing ventilator capacity by providing primary critical care support for multiple patient use in a cost-efficient device. The intuitive system combines sophisticated ventilation features with a plug-and-play setup to streamline patient care management. In addition, the system’s multi-stage antiviral filters are made to provide excellent infection control to protect both patients and healthcare providers.
Optimized for long-term storage with no maintenance, the reliable system eliminates the need for costly service contracts required for ICU ventilator systems, according to the company. COVID-19 transformed the market and RESPOND-19 aims to address the immediate and growing need for flexible, critical care ventilator capacity.
“Current critical care ventilators are not designed for periodic use, such as during global pandemics or disaster recovery. They require costly maintenance and additional time to deploy from long-term storage and are not optimized to prevent the transmission of respiratory illnesses,” says Richard S. Walsh, CEO of CorVent Medical.
“Our team’s expertise in ventilator development and proven track record of bringing innovative products to market positions us to successfully address this pressing clinical need. We are proud that the RESPOND-19 ventilator expands access to lifesaving respiratory support, in a financially sustainable way, for hospitals and government agencies during this healthcare crisis and beyond,” Walsh adds.
Featured image: RESPOND-19 Ventilator from CorVent Medical received FDA emergency use authorization (EUA) for primary critical care respiratory support.