
Christopher Harris, CBET, (R) mentors BMET Luong Nguyen (L) on the correct method to check a flexible GI videoscope.

L-R: Carl Bailey, Chris Botello (standing), and Travis Recksiek work to complete PMs on more than 3,800 infusion devices.
The history of Parkland Health and Hospital System (PHHS) has a rich past that began in 1872 when three local physicians opened the first permanent hospital in Dallas—in the midst of the city’s “red light” district—to care for indigent patients. Two years later, it opened a new hospital facility. This facility expanded over the next 20 years until a campus, paid for with voter-approved bonds, was built in 1894.
Development continued, expanding both Parkland’s facilities and capabilities over time. With each new advance, the institution implemented smart processes and procedures, becoming, multiple times, a model system.
In 1954, Parkland became the first Texas hospital to use a kidney dialysis machine. In 1955, Parkland was the site of the first corneal transplant. In 1956, it developed one of the nation’s first nuclear medicine labs. In 1962, the hospital began a nurse-directed triage program in its emergency department, another first for the nation. Over the next decades, the institution continued its innovative path: the first successful kidney transplant in Texas, the first high-risk maternity unit in the country, the first Level I trauma center in the state, the first pediatric trauma center in the nation.
Throughout the development, Parkland maintained its focus on patients in need. In 1872, the city hired a physician to care for prisoners. Today, Parkland continues to serve the Dallas County Jail Health System, including three adult jails, two juvenile facilities, and the county’s juvenile intervention camp. At the same time, the hospital has treated high-profile patients, including victims of the region’s 1957 tornado, survivors of the crash of Delta Flight 91, and, most famously, President John F. Kennedy, following his shooting in 1963.
“A big philosophy here is that we’re on a journey to excellence,” says Tom Collins, Parkland’s director of clinical engineering and biomedical services. That philosophy is guiding the institution’s next big move—into a new state-of-the-art facility in 2014 that the hospital says is currently the largest building project in the nation. “We want to have excellent processes in place in the new hospital, and anything we can do to improve now is embraced by the leadership of Parkland,” Collins says.
Better Management
As part of its effort to improve for the move, the Parkland clinical engineering team decided to take a look at the department’s policies for areas that could be enhanced. Some changes seemed obvious; others were subtler.
“Commonly, people who write policies like to use words that will trap you—’must,’ ‘will’—words that leave no room for flexibility,” says Don Allen, biomedical manager for the Parkland clinical engineering team. However, policies should not require processes that will not get done—nor should they be used as an enforcement tool. “That should be a managerial responsibility,” Allen says.

L-R: BMET Chris Botello completes a performance verification on defibrillators with senior BMET Abraham Mathew at the Dallas County Jail.
Offering an example, Allen describes how former policy stated that all equipment needed to come through the biomed department for inspection before delivery to the clinical site. “We don’t have room to receive a CT scanner, so we know that it cannot happen that way,” Allen says. Subsequently, the policy needed to be rewritten to permit changes when the department could not meet the desired process.
“We also looked at our PM procedures and frequencies and recognized that there was a great opportunity to bring them up-to-date,” Collins says. The team performed failure-based analyses on those devices to determine where they could justify fewer requirements. In some cases, procedures changed. “We were still using oscilloscopes to check outputs on ESUs,” Allen says. In others, the team increased the time between PMs.
The changes improved patient safety and at the same time they freed technicians, giving them time to focus on services more critical for the clinical departments. Still, the adjustments took some getting used to on the part of the biomed team. “We have several technicians that have been here for many years, 10 or more, and doing things like changing PM procedures can be extremely uncomfortable for them because they feel we’re putting our patients at risk,” Allen says.
Education (often in the form of trade articles) and time won them over as they saw the changes did improve quality and were not merely cost-saving managerial moves or a cover for staff reductions. There was similar discomfort, though of a different nature, with the change in service delivery.
Better Service
“When I first came on here just a year ago, I realized that on the biomed side, nursing was actually bringing equipment down to the biomed lab, and we felt very strongly that nurses had better things to do than spend their time bringing equipment down to our shop,” Collins says. So the department implemented a “rounding” policy, where biomeds perform as much work as possible, including PMs, on the floors.
“Our staff is very technically skilled, but as is often the case with biomeds, they’re not the most outgoing personalities, if you will, and some of them had a tough time having to go up on the floors,” Collins says. Unlike the changes to PMs, however, the improvements were obvious right away, primarily in turnaround.

L-R: Clinical Engineer Matt Wied and Radiology Engineer Ken Fairfax examine the gantry on a CT unit during a performance-verification check.

Clinical Engineering Director Tom Collins (C), discusses database issues with Clinical Engineering Radiology Manager Ed Smith (L), and Clinical Engineering Biomedical Manager Don Allen (R), on their electronic tablets in front of the new hospital construction site.
“The old way of doing business would involve bringing the equipment down to the biomed shop, performing a troubleshooting analysis, ordering the parts or taking the next necessary step, completing the repair, and calling the department to arrange pickup or delivery,” says Christopher Harris, CBET, a Parkland BMET II. “From beginning to end, that was a little bit longer than we wanted to have the equipment out of service.”
By visiting the floors with the goal to resolve the issue in real time, if possible, the turnaround became much shorter, which Harris estimates works in the majority of cases. “We can take care of about 70% of problems on the floor, and the other 30% that may need a little bit more extensive repair, we can still bring down to the biomed shop for the work,” Harris says.
As part of their rounding, the clinical engineering team has also been charged with noting the need for any other repairs, even if they fall outside of the defined biomed scope. Examples include stained ceiling tiles, malfunctioning outlets, spills for environmental services, or even an unplugged balloon pump. “If they see anything that they wouldn’t put their family next to, then we shouldn’t be putting patients next to it,” Allen says of the guiding philosophy.
Better ProcessesThe move to a rounding service delivery method has been successful but has not been without some challenges, documentation being a primary one. On the floors, biomeds would have to take notes on their work orders, then enter them into the system at the end of the day (assuming the notes could be read and deciphered), creating duplicative work.
The solution has been to invest in HP tablets from Hewlett-Packard in Palo Alto, Calif—an equipment delivery the team is looking forward to implementing. Administrative buy-in was equally easy to obtain. “The cost on the tablets was about a third of what a laptop costs, and a pilot program showed that being able to open and close work orders on the floor rather than having to write everything down and duplicate the process at the end of the day was going to pay off in the long run through fewer wasted man-hours,” says Ed Smith, the biomedical manager of the radiology team for the Parkland clinical engineering department.
Team members will use the tablets to not only handle work order documentation but also to access the clinical equipment management software, or CEMS, to research information, such as device history or part numbers. And, additional technologies are expected to bring further benefits.
“We’ll have bar code readers that can be used to scan device bar codes to find out exactly what is in front of you and whether a problem has been seen before,” Harris says. “We’re anticipating it will make troubleshooting a lot easier.” The use of active tags will help with real-time location of devices, particularly pumps. And there are thoughts that, eventually, Skype (or a similar system) could be used for communication purposes, such as remote technical assistance.
Knowing the current and future needs, the team was careful to make sure whichever tablets they selected would work for their purposes. “We went through roughly six different units from different manufacturers, even the iPad, and were limited by the applications we interface with,” Smith says. The experience taught the team and manufacturers that applications other than Internet Explorer are necessary to ensure interface capabilities with various health care software systems. The team settled on HP because it was able to run a “true Windows mobile-type application” and worked with the department’s CEMS from Four Rivers Software Systems, Pittsburgh.
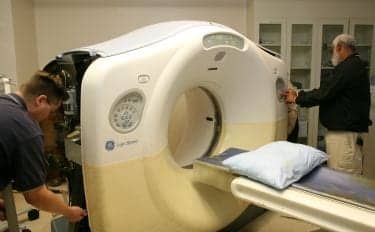
L-R: Radiology Engineers Scott Adams and Ken Fairfax begin a performance-verification check on a CT system.
Better Tools
In making the tablet selection, the team worked with the chief information officer (CIO) on selecting a product that would meet both IT and clinical engineering’s objectives. After the team presented evidence on its selection, the CIO agreed with the biomed department’s choice.
This type of process/presentation is often repeated during capital equipment acquisition, which is typically a multidisciplinary effort. Naturally, the biomedical department plays a large role.
“We are part of the team from the very beginning: the process of RFP, the site visits, the analysis of the equipment, and the pros and cons of each device and manufacturer,” says Bud Gunter, CT specialist at Parkland. “And, a lot of times, we can come up with insights that the department heads would not even think to ask about.”
The team’s advice focuses on the device’s impact in the shop but more on how it affects patient care and department operation. “Since we come in contact with equipment all the time, we try to be a resource of knowledge and experience for the clinical staff so that they can focus on patient care,” Harris says. “We can give them a concise breakdown of what we feel is the best equipment to fit their needs.”
That process has kicked into high gear with the planned move into the new facility. The 31-member clinical engineering department currently provides service and maintenance for approximately 17,000 devices within the hospital and 11 community health clinics, including the jail system. It is now faced with determining which equipment will be viable for the 2014 move and which devices will need to be replaced.
A computer generated mock up of a patient room in the new hospital.

The new 862-bed hospital is scheduled for completion in winter 2014.

The new $1.27 billion Parkland hospital will add desperately needed space for the better delivery of health care services to a growing population, particularly in critical areas such as the emergency department, operating room, burn center, and neonatal intensive care unit.
To ensure smart decision-making, the team recently created a new position, hiring clinical engineer Greg Stett to focus on equipment in the new facility. Stett will work with the existing hospital’s clinical engineer, Matt Wied, to select devices for the move and plan their relocation. Devices that are easy to transport, such as portable digital devices and C-arms, are having capital dollars allocated now; new purchases will include relocation costs when possible.
Items involved in intricate systems will require significant planning. “Relocating an x-ray machine is a challenge, but it’s easier to schedule moving around an x-ray machine than it is to schedule the move of an entire ICU,” Allen says. “Our challenge is, how do we continue to see and monitor patients in an active ICU and disconnect it and relocate it?”
Clinical teams have been directly involved in room layout design, and the hospital is incorporating philosophies from Lean and Six Sigma process-improvement methods. “At our new hospital center, called Cityville, there are mock rooms set up for clinical staff to visit and provide feedback,” Collins says. Comments can cover anything from where to place patient monitors to how the headwalls look.
Better Knowledge
Whatever decisions are made, the clinical engineering team will be prepared. Training is key to the department’s function and, in the past year, it saw its training budget more than double over the previous year. “The increase was scrutinized, but we were able to show that by taking additional equipment off contract and servicing it ourselves, we would be able to increase response time and decrease equipment downtime with tremendous cost savings—especially after the first year—through reductions in contracts,” Collins says.

L-R: Biomed Manager Don Allen discusses jail health issues with clinical engineering Director Tom Collins.
The team tries to handle as much service in-house as possible. Equipment for which vendors do not facilitate in-house service or for those where the risk or cost is not justified (as with clinical laboratory analyzers) will fall under contract, with everything else handled by the team.
To enable this, whenever possible, training is negotiated into a capital acquisition. “It is always part of the conversation when we talk to vendors,” Harris says. When training is not available through negotiation or is needed due to turnover, the department will pay for outside instruction.
Often, the department sends the biomed considered the primary responder for the equipment to training, with the expectation that he will share that knowledge when he returns. Using balloon pumps as an example, Harris explains, “You would have the primary technician trained at the vendor school, but you would also have a secondary guy who may be trained just from the primary tech.”
As the imaging team’s CT specialist, Gunter has been involved in much cross training. “I’ve got a number of CT schools coming up with secondary techs scheduled behind me, and once we have that training, we’ll come back and pass it on to other in-house engineers,” Gunter says.
Better Preparation
The collaboration takes place on a departmental level, with the biomed and imaging teams working closely together, a relationship that has evolved gradually over the years. Each group is comprised of 12 technicians or engineers and one manager; Collins, two clinical engineers, and two administrative staff round out the team.

The new Parkland hospital is currently the nation’s largest hospital project under construction.
“There’s a bigger focus on teamwork with present management,” Harris says. The result is a tight department that found it was able to handle its first Centers for Medicare and Medicaid Services, or CMS, inspection—which the team jokingly equates to a “Joint Commission inspection on steroids”—with relative ease.
Explaining the difference further, Allen notes that The Joint Commission inspections focus on outcomes: If problems are found, the facility can amend its policy and take appropriate corrective action. CMS inspections operate on a pass/fail basis, where if one flaw is found, that entire portion of the program will fail.
“It’s like they take a snapshot of your operation to see if you passed or failed on each key or regulation that they are looking at,” Collins says.
This meant that the CMS inspection team asked for much more documentation than is usually presented to The Joint Commission. Requests came in on a daily basis during the 2-week audit; questions and discrepancies were addressed in meetings. “They questioned our documentation and why we did things a certain way,” Collins says. “We were able to justify our actions, and they were fine with that, but it was a more thorough and deeper survey than it would have been with The Joint Commission.”
Despite the additional work, the inspection did have one key benefit: “It verified for staff that everything we’ve been talking about over the previous 6 to 12 months paid off,” Collins says. And it showed that the department and the institution continue on their journey to excellence.
Renee Diiulio is a contributing writer for 24×7. For more information, contact .




