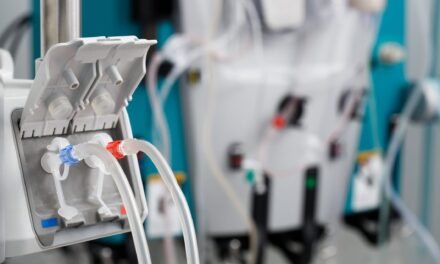
Smiths Medical is recalling Medfusion 3500 and 4000 Syringe Infusion Pumps for eight software malfunctions that affect different serial numbers and software versions.
These malfunctions may cause serious harm or death to patients from under- or over-infusion, or delays in the delivery of critical medications to patients. The FDA has identified this as a Class I recall, the most serious type of recall. So far, 118,055 devices have been recalled in the U.S.
Smiths Medical Medfusion 4000 and 3500 Syringe Infusion Pumps are used to give fluids to patients in precisely controlled amounts. They deliver blood or blood products, lipids, drugs, antibiotics, enteral feedings and other therapeutic fluids through infusion tubing into a patient’s vein or through other cleared routes of administration. Syringe pumps are primarily used in the neonatal and pediatric populations or in operating rooms and intensive care units for the adult population.
The eight software issues include:
- False alarm for primary audible alarm system failure.
- Unanticipated depleted battery alarms.
- Abnormal circuit board behavior, which may cause internal clock system failure.
- Intermittent volume over time delivery mode where the infusion continues after system failure.
- Unanticipated clearance of program volume delivered.
- False alarm for rate below recommended minimum for syringe size.
- Incorrect bolus or loading dose time display.
- Network configuration may affect pump communications.
Smiths Medical states there have been a total of seven serious injuries and one death reported related to these issues. The Customer NotificationExternal Link Disclaimer identifies the injuries and/or deaths associated with each software issue.
Featured image: The Medfusion 4000 Syringe pump from Smiths Medical