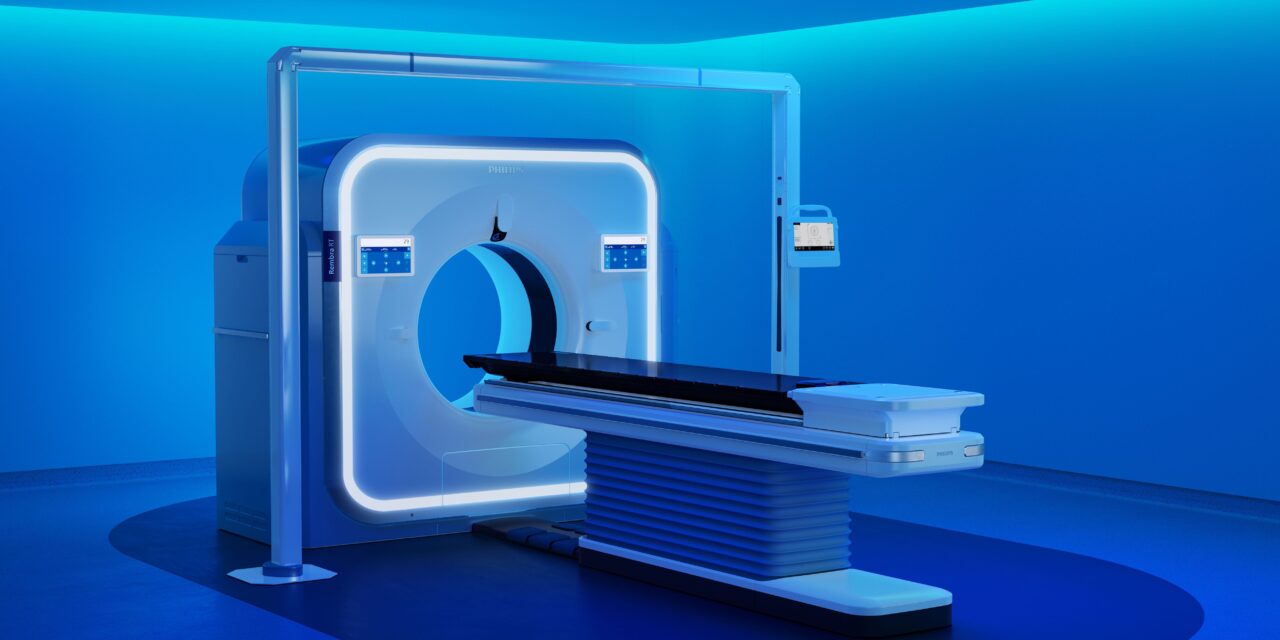
Clearances for Rembra CT, Rembra RT, and Areta RT aim to enable high-throughput imaging at scale and elevate precision across the cancer care pathway.
Royal Philips announced it has received 510(k) clearance from the US Food and Drug Administration (FDA) for its Rembra platform of scanning systems: Rembra CT, Rembra RT, and Areta RT.
The newly cleared systems are intended to help healthcare providers address growing demand for imaging and radiation therapy planning by supporting speed, efficiency, and access to imaging, Philips says in a release. The company notes that increasing imaging volumes and clinical complexity are driving the need for systems that can support workflow efficiency while maintaining diagnostic confidence.
According to Philips, the Rembra platform is designed to extend the role of CT across both diagnostic radiology and radiation therapy, supporting more connected workflows across care settings, from acute imaging to cancer treatment planning.
“As healthcare systems manage increasing demand and complexity, imaging plays a critical role in enabling timely and informed clinical decisions,” says Dan Xu, business leader of CT at Philips, in the release. “With the Rembra platform, we are redefining what clinicians can expect from CT, combining speed, scalability, and precision to expand access to high-quality imaging while supporting confident diagnosis and highly accurate treatment planning.”
Philips says Rembra CT features an 85 cm bore and is designed for high-throughput imaging in settings such as emergency departments, critical care, and interventional environments. The company reports the system can support up to 270 exams per day based on internal testing assumptions. It also includes acquisition and reconstruction capabilities intended to support rapid imaging.
The Rembra RT and Areta RT systems are designed for radiation therapy planning and include features such as a wide 85 cm extended field of view and 4DCT imaging capabilities, according to the release. Philips says these systems are intended to support tumor targeting while limiting exposure to surrounding tissue, as well as streamline simulation and planning workflows.
Philips describes the Rembra platform as a unified CT system spanning diagnostic imaging and radiation therapy planning. The company says the platform is intended to help providers manage imaging demand while supporting more consistent workflows across care settings.
The FDA clearances add to Philips’ CT portfolio and reflect its focus on integrating imaging systems with connected workflows, according to the company. Philips says the platform is part of its broader effort to align diagnostic imaging and therapy planning technologies.
Photo caption: Rembra RT
Photo credit: Philips