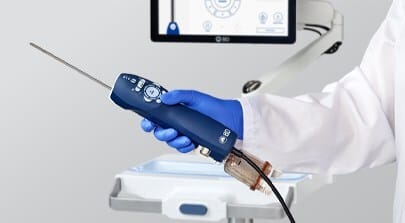
ALung Technologies, Inc., a provider of low-flow extracorporeal carbon dioxide removal (ECCO2R) technologies for treating patients with acute respiratory failure, announces that the U.S. FDA has granted the company De Novo clearance for the Hemolung Respiratory Assist System.
The Hemolung is indicated for respiratory support that provides extracorporeal carbon dioxide (CO2) removal from the patient’s blood for up to five days in adults with acute, reversible respiratory failure for whom ventilation of CO2 cannot be adequately or safely achieved using other available treatment options and continued clinical deterioration is expected.
“The FDA De Novo review process is designed to determine that the clinical benefits outweigh the clinical risks associated with a medical device when there is not a substantially equivalent predicate device,” says Peter M. DeComo, ALung’s chairman and CEO. “As a result, the De Novo process is quite rigorous.”
He adds, “In order to demonstrate benefit over risk, ALung submitted data to FDA from over 1,000 Hemolung patient treatments on clinical safety and over 230 Hemolung patient treatments on clinical performance outcomes. This clinical data along with all of our pre-clinical data demonstrated to the FDA that the clinical benefits of the Hemolung for ECCO2R therapy has been substantiated.”
The Hemolung is an all-in-one system intended to provide minimally invasive, low-flow ECCO2R. Low-flow ECCO2R with the Hemolung is a lung-independent ventilatory support therapy for removal of CO2 waste molecules from venous blood via extracorporeal circulation through a single, 15.5 French, central venous catheter at blood flows of 350-550 mL/min. Respiratory failure patients often experience the need for CO2 removal without the need for supplemental oxygen.
Historically, when extracorporeal therapy was indicated for the removal of CO2,extracorporeal membrane oxygenation (ECMO) devices were utilized because other alternatives were not available. ECMO is typically necessary when respiratory failure patients have a significant oxygenation issue. ECMO systems are complex, invasive, require high blood flows and as a result, require specialty personnel to monitor both the technology and the patient. The Hemolung offers ICU physicians, nurses, respiratory therapists and perfusionists a new tool to treat respiratory failure patients in a less-invasive manner, company officials say.