The FDA has awarded 510(k) clearance to Philips Healthcare’s line of CT products for low-dose lung cancer screening. The product line includes 27 CT and PET/CT models, as well as integrated software and services that work to detect lung cancer—the number one cancer killer in the United States.
Philips’ CT product line gives healthcare organizations of varying clinical and economic needs—from community hospitals to multifacility health systems—the ability to build lung cancer screening programs. Through its integrated radiology services, Philips can work with healthcare professionals to assess the availability of existing scanners and to establish an enterprise-wide lung cancer program.
“Early detection is key to fighting this terrible disease,” says Brady McKee, MD, a radiologist at Burlington, Massachussetts-based Lahey Hospital and Medical Center, which is currently conducting the nation’s largest clinical lung cancer screening program. “The work Philips has done to improve a provider’s screening capability not only benefits individual patients, but hospitals at each step of their lung cancer screening program implementation and execution, which is often a very complicated process.”
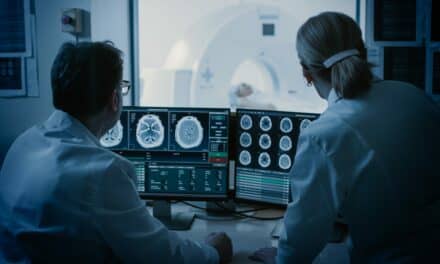
The manufacturer’s lung cancer screening products also provide several benefits for patients and referring physicians, including personalized management of radiation dose and image data-sharing and analytics. The IntelliSpace Portal facilitates the latter benefit—with the latest version of the portal including a CT Lung Nodule Assessment application designed to provide additional clinical decision support.
For more information about the product line, visit Philips Healthcare.