By Binseng Wang and Torgeir Rui
Senior hospital leaders often ask how long medical equipment can be used before replacement is needed, as they constantly struggle to secure investment capital for it. Many consultants use the age of the equipment as the primary, if not the only, parameter to determine the need for replacement. Some original equipment manufacturers, or OEMs, publish “end of life” (EOL) or “end of support” (EOS) notices to encourage hospitals to replace their equipment at a certain point in time.
One could contend that the age of the equipment should not be the only or even the best criterion to make this determination, because many other factors are likely to be just as important. As an analogy, the same brand and model of vehicle can be used by a full-time remote worker who only goes out for groceries and occasional social events, or by a ride-sharing driver trying to support his or her family by taking as many trips as possible. The first vehicle is likely to last much longer than the latter if both are put into service at the same time.
While such arguments make intuitive sense, someone could counter with the fact that while mechanical and pneumatic machines typically exhibit wear-out pattern (in the case of most vehicles), medical equipment nowadays is mostly composed of electronic components and, thus, can last many years without exhibiting clear signs of wear and tear. For example, the Hubble Space Telescope has been in operation since 1990!
The key criteria for medical equipment replacement should be a combination of safety, physical condition, supportability, and clinical impact. As clinical engineering/healthcare technology management (CE/HTM) professionals, we can affect the first three criteria. By performing safety and performance inspections, we can detect and solve hidden and potential failures that are unknown to the users or are in progress and, thus, we can enhance patient safety.
Through timely replacement of wearable parts (i.e., preventive maintenance), we can reduce predictable wear and, thus, boost the reliability and longevity of a device. Equipment availability is just as important as safety, as it has a direct impact on the timeliness of patient care, even to the extreme of denial of care if there is extended downtime. By utilizing service expertise and replacement parts—including alternative sources after the EOL/EOS declarations—we can ensure the availability of equipment.
If we complete all the tasks summarized above, can we prove that we are able to prolong the lifespan of medical equipment? To answer this question, we selected a subset of the medical equipment that Sodexo HTM has been managing (roughly 520,000 pieces of equipment) over the past three decades for its clients for which we have high-quality data, and performed an analysis of the reliability as a function of the equipment age.
Our results show some types of equipment exhibited clear wear-out patterns, whereas some did not, and still others were inconclusive due to insufficient data. At the same time, the data allowed visualization of how long equipment was deployed in those healthcare delivery organizations (HDOs).
Figure 1 compares our findings with the “estimated useful lives” (EULs) published by the American Hospital Association (AHA). It is evident that the equipment types that we have examined to date are used for much longer by HDOs than the EULs suggested by AHA for both biomedical and imaging equipment (indicated by B and I preceding the numbers) before they are replaced.
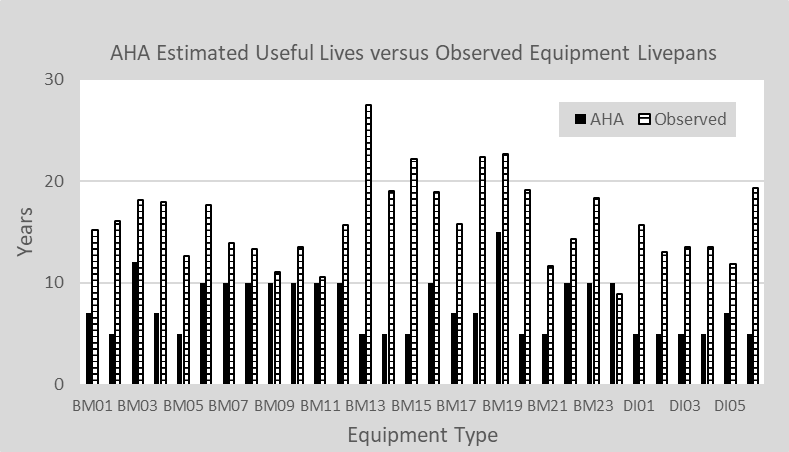
Figure 1: A comparison of the estimated useful lives provided by AHA and those observed in Sodexo HTM client facilities.
This finding by itself does not unequivocally prove that we are successful in prolonging the equipment’s lifespan for several reasons. First, the AHA cautions readers that “[t]he estimated service life for each asset as presented in this booklet is to be used primarily as a guide. An organization may consider assigning a longer or shorter life depending on usage, types of facility, and extenuating circumstances affecting the service life of the asset.”
Second, AHA acknowledges that equipment replacement is often motivated not solely by deterioration, loss of reliability, or high maintenance cost, but also by “…technological innovation, which can render an asset obsolete before the end of its estimated useful life.” Finally, it seems that AHA EULs are more often used for financial depreciation calculations than actual replacement evaluations, as it states in the booklet that “[t]he method for determining the depreciable cost is largely dependent on the productive period of the asset. Numerous factors influence this determination… “ (CMS requires the use of AHA EULs for Medicare/Medicaid program participants in their capital depreciation calculations).
On the other hand, it is clear that the age of the equipment alone should not be used to make decisions about equipment replacement. Likewise, the EOL/EOS declarations by respective OEMs should be taken with a “grain of salt,” considering the inherent conflict of interest.
Despite those limitations, we believe each CE/HTM team should analyze and share similar data with their respective senior hospital leaders. In doing so it is likely that a case can be made that if given proper support and resources, CE/HTM professionals can help HDOs to confidently utilize medical equipment for many more years than those suggested by AHA, some OEMs, and consultants.
In terms of the financial impact of prolonging the useful lives of equipment, a very gross estimate can be obtained by using the current estimated annual capital investment in medical equipment by all community (i.e., non-government or military) hospitals, i.e., approximately $24 billion per year[1].
If equipment replacements had to be accelerated to match the AHA EULs, one would have to more than double the current capital equipment investment (data shown in figure 1 indicate that actual deployment time is on average 2.3 times the AHA EULs). In other words, community HDOs would have to find an additional $31 billion of capital investment per year, totaling around $55 billion, which is a number that is virtually impossible to reach in normal years, much less after the serious financial crisis caused by the COVID-19 pandemic.
Binseng Wang, ScD, CCE, fAIMBE, fACCE, is vice president of program management at Sodexo HTM. Torgeir Rui is lead data analyst of program management at Sodexo Healthcare Technology Management. Questions and comments can be directed to 24×7 Magazine chief editor Keri Forsythe-Stephens at [email protected].
[1] See “Comment from Sodexo Clinical Technology Management LLC” available at https://www.regulations.gov/comment/FDA-2021-N-0561-0013






Important article-where is Figure 1?