SECO USA, a subsidiary of SECO SpA, a manufacturer of computer miniaturization and “ready-to-use” Internet of Things integrated systems, announces that multiple Biorespira Pulmonary Ventilators will be donated to hospitals in the U.S., with priority given to Black communities.
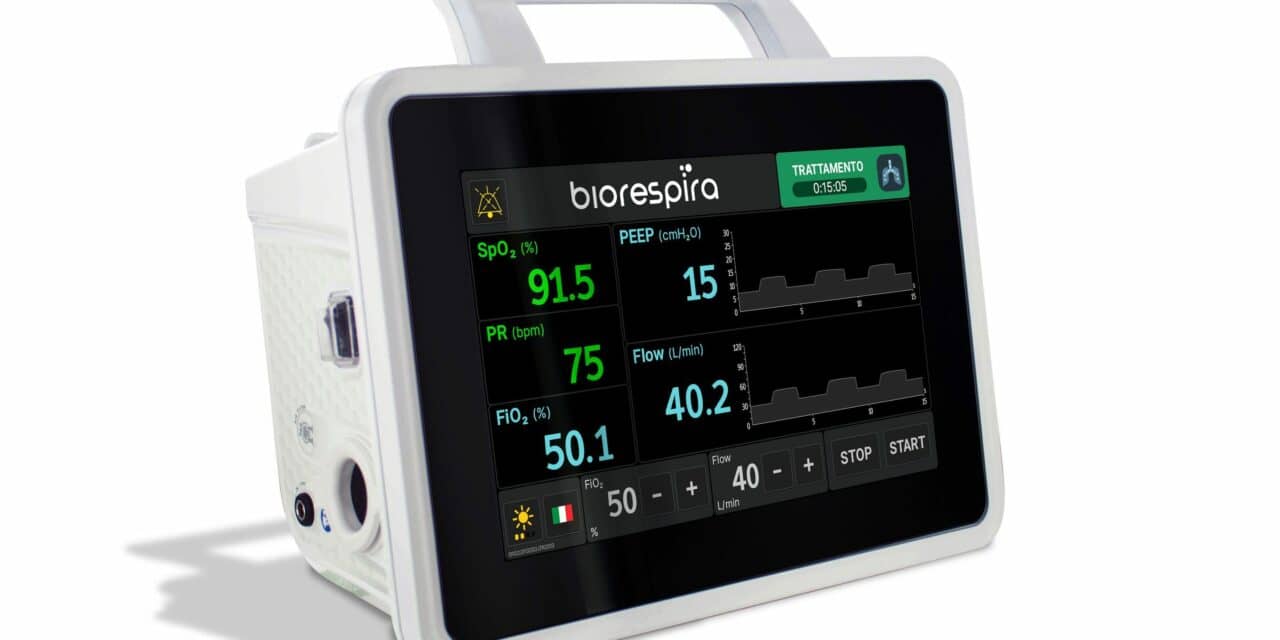
Biorespira is a non-invasive pulmonary ventilator that delivers oxygen to patients with difficulty breathing in a closed loop system, easily operated by nurses and in-home care providers. By prioritizing the donation of Biorespira Pulmonary Ventilators to facilities in Black communities, SECO USA hopes to reach people who too often have to wait for the most effective healthcare in times of crisis.
Biorespira provides healthcare providers with a device designed to oxygenate adult patients without intubation in almost any location—outside of an ICU, in hospitals’ general medical wards, nursing homes or private residences. Due to the simplicity of the interface, nurses, in-home care providers and family members require only minimal training to operate Biorespira.
Biorespira was created as a high-flow system, providing an adjustable percentage of oxygen from 21% to 100% and a flow rate between 10 to 120 L/min. Using medical helmets or a non-vented face mask with Biorespira ensures a closed loop system, nearly eliminating the chance of the SARS-CoV-2 virus that causes COVID-19 passing from patient to caregiver.
Driven by the desperate need for ventilators during the early stages of the COVID-19 epidemic in Italy, Italian Biomedical Devices S.r.L, a local technology company led by Corrado Ghidini, MD, DDS, MBA, quickly created the noninvasive and portable ventilator, going from concept design to first prototype build in just five weeks. An existing relationship with SECO SpA evolved to include the distribution of Biorespira worldwide.
“At SECO USA, we feel it is vital to make the life-saving capabilities of Biorespira available to people in Black communities in the United States,” says Greg Nicoloso, CEO for SECO USA. “SECO USA has partnered with IBD to bring the Biorespira Pulmonary Ventilator to the U.S. market—our goal with this donation program is to deliver this vital capability to under-resourced hotspots in time to reduce deaths from COVID-19.”
The pre-EUA application for the Biorespira Pulmonary Ventilator has been submitted to the FDA. Units will be available in early July, once approved by the FDA through EUA or other certification.