
David Braeutigam
You have probably walked by them before, perhaps without even realizing it. They are in airports, hospital corridors, government buildings, and schools. And despite their critical importance in a time of sudden cardiac arrest, automated external defibrillators (AEDs) can expire without anyone taking notice, rendering them useless when their functionality is needed most.
Sold by numerous manufacturers, AEDs have become an integral part of public safety planning. Unfortunately, without proper monitoring and maintenance, the devices—notably, the battery and electrode pads that both have expiration dates that are often ignored by owners—can become obsolete. While manufacturers include educational materials and safety alerts to warn owners of these risks, there is little or no quality assurance by manufacturers or governing bodies, placing the burden of maintenance entirely on the owner—someone who may or may not understand the importance and upkeep of critical medical devices.
Widespread Concerns
“We have distributed over 800,000 AEDs worldwide since about 1996,” says Jamie Froman, Philips HeartStart AED global director of marketing. “We’re coming up on almost a million devices distributed from Philips alone. There are a pretty substantial number of devices out there.”
With so many AEDs in the market, policing them is a difficult task for any governing body, let alone the manufacturers that make the devices. In an effort to mitigate these risks, Philips implements several warning signals and precautionary notices in its HeartStart AEDs, including instructional manuals, self-test capabilities, and visual and audible alert systems.
“Device readiness is one of the key things that we monitor,” Froman says. “These devices will spend many days on a wall, standing by and ready for use. Then, there’s one day when all of a sudden, they’re needed. We want them to be ready at the moment when someone has had a sudden cardiac arrest and needs the device. There’s a lot of engineering and thought that goes into what we call the ‘self-test’ area.
“When the self test identifies something that might be wrong in the future, the system will start chirping at you, the green status light goes from blinking to solid green, and the device will start calling attention to itself. People that are passing by will want to engage with the device and press the blue diagnostic button. Then the device will give a self diagnostic of what needs to be addressed on the device.”
But David W. Braeutigam, MBA, CBET, director of biomedical engineering, Baylor Health Care System, Dallas, warns that identifying and resolving AED issues may not be quite that simple. Braeutigam recently visited a car dealership that displayed an AED for urgent care and, upon checking the pads and battery, found that not only were the pads expired by 2 years, but the battery was completely dead. A week prior, a foundation donor contacted Braeutigam’s hospital after the local fire department discovered that the AEDs in the donor’s residence were expired.
“I don’t think these are isolated incidents,” Braeutigam says. “The general public does not know that the pads expire after a couple of years or that the batteries expire after 4 to 5 years.”
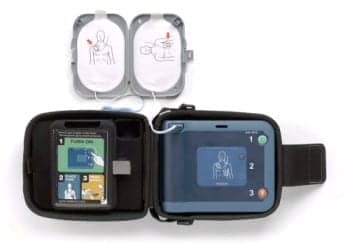
Battery and electrode pad expiration dates are often ignored by owners.
This obviously raises several questions: Are the safety precautions that manufacturers implement actually working? If not, is there more that they can do? And are there any governing bodies that should monitor these devices?
The FDA currently clears AEDs through the 510(k) regulatory process and lists them as Class III medical devices, according to the 1976 Medical Device Amendments. However, according to the FDA Web site,1 after a 1990 amendment to the 1976 legislation, “The FDA must either down-classify AEDs to Class II or keep AEDs as Class III and require they go through the more stringent premarket approval process. The FDA is now proceeding with the formal classification of AEDs.” In January 2011, the FDA convened an advisory panel meeting to discuss these regulatory pathways and which would be best for AEDs to provide reasonable assurances of safety and effectiveness.
This classification concerns only the devices’ premarket approval, though, and does not concern the upkeep of them in public or medical settings. So while Froman believes that most manufacturers have a vested interest in the quality assurance and upkeep of their AEDs—likely necessitating similar warning features and educational information—there are no regulations regarding the maintenance of AEDs.
Ultimately, the responsibility for the devices’ functionality falls on the owners, but even with safety alerts already in place, a very real problem exists. Despite that, Froman says that not only is it difficult to ignore or turn off these alerts, but Philips offers the option for customers to receive notices about expiring parts.
“We’re always balancing the ease of use and safety aspects,” Froman says. “We need the device to be easy to use in the moment. However, when the device needs attention, we need the person that’s hearing that chirping—it’s a pretty loud, persistent chirp, a bit like a smoke alarm—to engage with the device.
“One of the things that we offer customers is the option to purchase an online service that we call SMART Track, which helps keep track of the expiration date on their pads and batteries. The program manager for the IT program would work with Philips to load their dates for supplies into the SMART Track system, and the application will automatically notify an owner via e-mail 90 days prior to a part’s expiration date. It will send repeat e-mails until the person indicates that they’ve replaced the supplies.”
Unfortunately, as Braeutigam’s experiences demonstrate, there is a disconnect between the established safety alerts and the owners of AEDs. One issue that he mentions is the placement of these devices in areas that attract little or no traffic, causing the alerts to go unnoticed.
“The dealership I went to had three AEDs, and they are mounted in cabinets. You’re going to walk by them every day, but you’re not going to look in the cabinet to see if the light is on,” Braeutigam says. “If you’re not walking by it when it’s beeping, you’re not going to hear it.”
Fixing the Problem
“Somebody has to be involved, but I don’t know who it should be,” Braeutigam says, when asked about whether or not the FDA should play a more prominent role in the policing of AEDs in the public setting. One suggestion he offers is to ask local fire departments to add AED pads and batteries to a checklist used during regular inspections of public buildings. Asking biomeds to take a more proactive approach to the problem may also mitigate some of these concerns.
“We can ask questions of the owners of the AEDs,” Braeutigam says. “If you see one at your local auto dealer, your local school, your library, etc, ask someone if you could help them inspect it for expired pads and batteries.”
The lack of proper, continued functionality of AEDs in public settings is an ongoing problem that often goes unchecked, but is of prime importance. Though he doesn’t have any statistics, Braeutigam’s anecdotal evidence likely signifies a broader trend that needs to be addressed first by AED owners, but also by those in a position to help.
“There are a lot of public buildings where I feel like everyone has a sense of security because we have these AEDs,” Braeutigam says. “But, there’s probably a lot of them that, based on how long they’ve been around now, don’t work. They might, but I think that if someone did a thorough study of them, there would be a high percentage that do not work at all.”
Chris Gaerig is the associate editor of 24×7. Contact him at .
Reference
- US Food and Drug Administration. External Defibrillator Improvement Initiative. Available at: www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/CardiovascularDevices/ExternalDefibrillators/ucm232302.htm. Accessed March 15, 2011.





