Eitan Medical, the manufacturer of the Sapphire Infusion System, says it will now service customers of ICU Medical in France, U.K. and U.S. Eitan Medical will become the exclusive supplier and service provider for its own Sapphire Infusion System, including the provision of administration sets, accessories, spare and technical parts and repair and preventative maintenance services.
Eitan Medical will also serve as a distributor for administration sets manufactured by ICU Medical for the Sapphire Infusion System—explaining it will bolster its current administration set supply around the world.
“This transition marks the completion of a successful and beneficial partnership between Eitan Medical and ICU Medical,” says Igal Shany, CEO of Eitan Medical. “We are appreciative and thankful to Dan Woolson and the entire ICU Medical team for serving as a valuable strategic partner to Eitan Medical, and for working collaboratively to provide advanced infusion therapy solutions these past few years.”
Woolson, who serves as corporate vice-president and GM of ICU Medical, says he is excited about the partnership. “We look forward to the successful transition of the business back to Eitan Medical and ICU Medical’s ability to focus on our Best in KLAS Plum platform and ICU Medical MedNet software to improve the safety, efficiency, and operating effectiveness of our customers’ IV therapy practices.”
Shany echoes those sentiments. “Eitan Medical strives to enhance its commitment to patient care, comfort and safety through innovation and exceptional customer experience. We continue to illustrate our patient-centricity through continued development of our flagship Sapphire infusion pump, a proven and well-known solution within the healthcare ecosystem, capable of answering the evolving needs of patients in need via intuitive and patient-minded performance.”
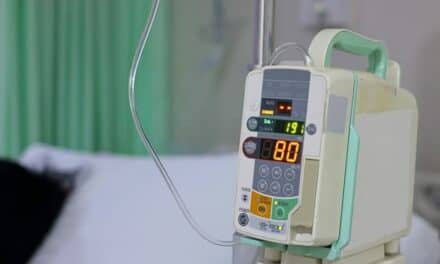
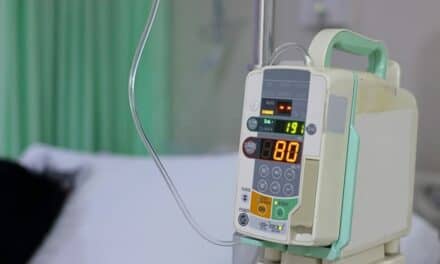
Featured image: Eitan Medical’s Sapphire Infusion Multi-Therapy Pump. Eitan Medical will be the exclusive provider of its Sapphire Infusion System for ICU Medical in France, U.K. and U.S.