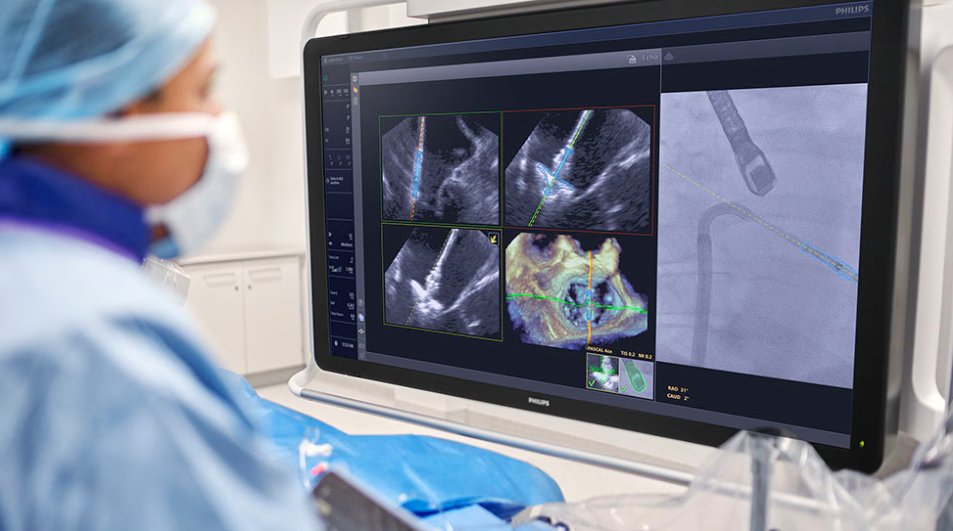
EchoNavigator with DeviceGuide uses AI to track and visualize repair devices in real time during minimally invasive procedures.
Philips has received US Food and Drug Administration (FDA) 510(k) clearance for EchoNavigator R5.0 with DeviceGuide, an AI-powered software solution designed to assist clinicians during minimally invasive mitral valve repair procedures.
The software is intended to support mitral transcatheter edge-to-edge repair, a procedure used to treat mitral regurgitation, a condition that affects more than 35 million people worldwide.
DeviceGuide integrates with Philips’ Azurion image-guided therapy platform and uses AI to track and visualize repair devices in real time. The system combines live echocardiography and X-ray imaging into a single view to support navigation and positioning during procedures.
Real-time Imaging and Guidance
Built on Philips’ EchoNavigator echo-fluoro fusion technology, the software combines live ultrasound images from the EPIQ CVxi system with X-ray imaging from the Azurion platform.
The AI algorithm tracks the Edwards PASCAL Ace mitral valve repair device and provides a combined visualization to help clinicians guide placement.
“The AI software serves as an assistive tool; the physician always remains in control. This isn’t about replacing expertise; it’s about amplifying it,” says Atul Gupta, MD, chief medical officer of diagnosis and treatment at Philips, in a release. “By embedding AI into the procedure, DeviceGuide gives physicians an extra pair of eyes, helping them treat more patients safely and confidently.”
Clinical Collaboration and Workflow Impact
The software was developed in collaboration with Edwards Lifesciences and evaluated with clinical partners in the US and Europe, including teams at NewYork-Presbyterian/Columbia University Irving Medical Center.
“Since AI auto-aligns imaging to the device in real time and continuously informs the interventionalist about the location of the device in space on the imaging screen, it minimizes unnecessary repositioning of the imaging window, streamlines procedural guidance, and may improve the precision of device implantation,” says Rebecca T. Hahn, MD, director of interventional echocardiography, in a release.
Susheel Kumar Kodali, MD, interventional cardiologist, adds in a release, “DeviceGuide provides me with a visual overlay, trajectory line, and orientation line of the therapy device in both live 3D echo and fluoroscopic images during mitral valve repair procedures. Having a single, intuitive presentation of real-time target, orientation, and auto device-aligned views simplifies this procedure and improves team communication.”
Availability
DeviceGuide enabled by EchoNavigator is FDA 510(k) cleared in the United States. Availability is subject to market release and regulatory requirements, and the software is currently intended for use with the Edwards PASCAL Ace Mitral Valve Repair System.
Philips plans to showcase the technology at the American College of Cardiology 2026 meeting.