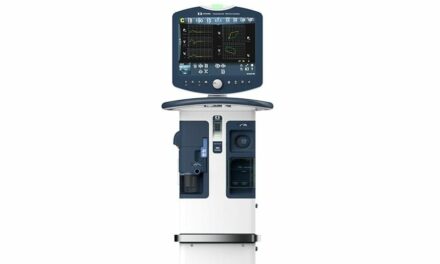
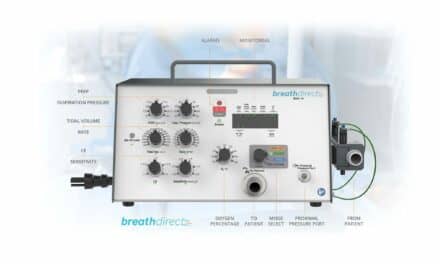
Vent Multiplexor, LLC and Yale New Haven Hospital announce that the U.S. FDA has granted Emergency Use Authorization to the Vent Multiplexor, an emergency rescue device they co-developed. The Vent Multiplexor is a patent-pending device designed to provide individualized emergency crisis care for two adult patients requiring mechanical ventilation when limited to a single mechanical ventilator.
The FDA Emergency Use Authorization comes just over a week after Yale New Haven Hospital successfully deployed the Vent Multiplexor to co-ventilate two critically ill COVID-19 patients with different disease states, different lung sizes, and different tidal volume requirements, all while accurately measuring lung compliance of both patients in real time.
Vent Multiplexor President Todd Higgins spoke out about this achievement, stating: “When the history of the COVID-19 pandemic is finally written, there will be a long list of heroes to thank. Included among them will be the dedicated public servants at the FDA quietly doing our nation’s work in partnership with the private sector to ensure that life-saving innovation quickly finds its way to the frontline.”
With FDA authorization in place, Vent Multiplexor, LLC, will ramp up production while also beginning to work with partners in countries where looming medical equipment shortages threaten a wave of humanitarian crises. After all, Higgins says, “Ultimately, we are all part of a global community. The call to action is for all of us to do everything in our power to help wherever that help is needed. We intend to answer the call.”
Thomas Balcezak, MD, Yale New Haven Health’s chief clinical officer and EVP welcomed the FDA authorization. “Yale New Haven Hospital is proud to have collaborated with Vent Multiplexor, LLC, and other groups on the development and successful trial of new technologies. We are committed to doing everything in our power to bring urgently needed new technologies and treatments to the community in the fight against COVID-19.”