Sweden-based Getinge announces that it has received clearance from the U.S. FDA for several new software options for its Servo-u and Servo-n ventilators. In addition to the latest software upgrades, Getinge has also received clearance for the new Servo-u MR ventilator for the MRI room.
“The COVID-19 pandemic and the heightened awareness of respiratory health has driven the need for personalized ventilation solutions for critically ill patients,” says Eric Honroth, president, Getinge North America. “Now more than ever, options for personalized lung protection and personalized weaning solutions are at the forefront of respiratory patient health. Getinge strives to support clinicians and patients by optimizing lung protection and delivering solutions for personalized ventilation.”
With this software upgrade for the Servo-u and Servo-n combined, Getinge adds several new functionalities and options across all patient categories—adult, pediatric, and neonatal. Moreover, Getinge broadens its portfolio of lung-protective tools, including Automatic Stepwise Recruitment maneuver, a standardized and automated workflow that guides lung recruitment and helps clinicians identify a personalized PEEP that provides the lowest driving pressure.
Further, stress index and transpulmonary pressure monitoring, including key parameters for assessment of lung stress during controlled and spontaneous ventilation, complements the lung protective toolkit, which was designed to divide the cognitive workload between the clinician and the ventilator.
Additionally, the clearance includes Heliox therapy, Getinge officials say. Heliox is a mixture of helium and oxygen that facilitates laminar flow and minimizes airway pressure due to its low density. This helps reduce the work of breathing of patients suffering from obstructive lung diseases.
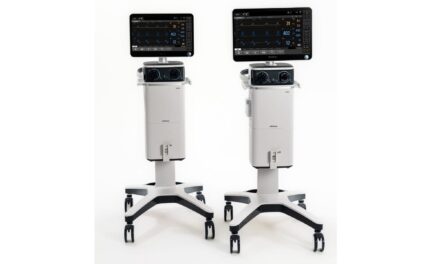
Finally, Getinge has also received clearance to introduce the Servo-u MR to the U.S. market. Designed to guide the ventilator into a safe position, the Servo-u MR includes a magnetic field indicator with visual and audible alerts and an auto-lock handle that locks all four wheels as soon as the clinician releases the ventilator.
“We are seeing a transformation in the way healthcare providers view respiratory health,” says Eric Honroth. “With this clearance, we are excited to be part of driving this transformation, working hand in hand with experts and clinicians.”
The new options and the Servo-u MR ventilator are expected to be available in the U.S. in July 2021.