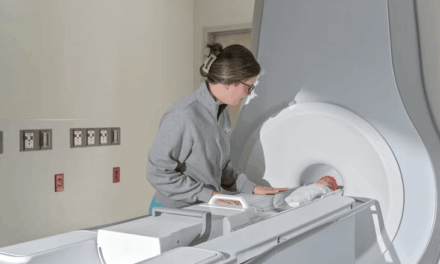
The 5mm Saberscope features a 90-degree articulating tip and integrated camera designed to reduce fogging and eliminate reprocessing needs.
The Food and Drug Administration (FDA) has cleared Xenocor’s Saberscope, a single-use 5mm articulating laparoscope designed for minimally invasive abdominal and thoracic surgical procedures.
The device features a single-use camera integrated into a 5mm rigid shaft with a 90-degree articulating tip, paired with a compact reusable video processor. The system is cleared for diagnostic and therapeutic endoscopic procedures within the thoracic and peritoneal cavities, including procedures involving female reproductive organs.
Traditional laparoscopic procedures rely on expensive image processing systems and reusable surgical scopes that require cleaning and reprocessing between patients, according to a release from the company, which notes that these systems can require hundreds of thousands of dollars in capital investment, while scope reprocessing introduces risks of cross-contamination and procedural delays.
“What stands out about the Saberscope is the reliability,” says John Langell, MD, Cleveland, Ohio, in a release. “Every case begins with a new scope that delivers a clear, consistent image without concerns about contamination, damage, or performance degradation.”
Designed to Reduce Fogging and Equipment Delays
The Saberscope system connects via universal plug-and-play functionality and displays images on standard HD monitors or through existing hospital tower systems. After each procedure, clinicians dispose of the scope to reduce biohazard exposure and ensure each case begins with new, non-contaminated equipment.
Clinical use and company test data show the technology reduces fogging during procedures, which can help maintain image clarity and limit procedural disruptions.
“The steerable tip fundamentally changes what I can see,” says Langell in a release. “It allows me to achieve unprecedented views of the anatomy while maintaining a clear, consistent image throughout the case. That level of visualization—without fogging, interruptions, or scope exchanges—improves efficiency and supports safer, more controlled surgery.”
The system aims to eliminate maintenance requirements and large upfront capital expenditures while reducing risks of burns and arc injuries associated with reusable scopes, according to the company.
“By combining a single-use articulating scope with a compact reusable video processor, we help eliminate fogging, reduce the risk of burns and arc injuries associated with reusable scopes, and avoid delays caused by unavailable or malfunctioning equipment,” says Tony Watson, chief operating officer of Xenocor, in a release.
ID 264994138 © Andrei ASKIRKA | Dreamstime.com