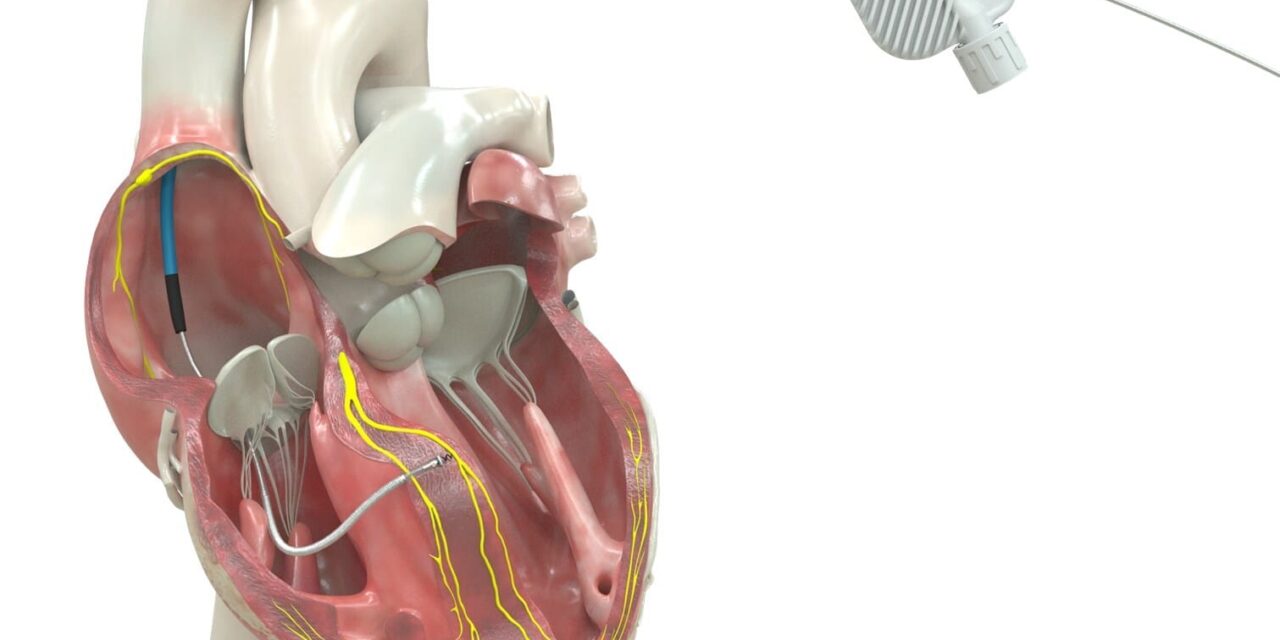
The defibrillation lead, approved for placement in the left bundle branch area, can deliver both life-saving shock therapy and physiologic pacing through the heart’s natural electrical system.
Medtronic has received Food and Drug Administration (FDA) approval for an expanded indication for its OmniaSecure defibrillation lead, marking the first time a defibrillation lead has been cleared for placement in the left bundle branch (LBB) area, according to the company.
The expanded approval enables the lead to be used for conduction system pacing (CSP), a method that closely mimics the heart’s natural electrical physiology and may reduce complications associated with traditional pacing approaches.
The OmniaSecure lead connects to an implantable cardioverter-defibrillator (ICD) or cardiac resynchronization therapy defibrillator (CRT-D) to treat ventricular tachyarrhythmias, ventricular fibrillation (VT/VF), and bradyarrhythmias. Beyond its new LBB area indication, the lead holds two additional distinctions: It is the smallest-diameter defibrillation lead on the market at 4.7 French (1.66 mm), and it is the only defibrillation lead approved for use in both adults and adolescent pediatric patients ages 12 and older, according to a release from Medtronic.
Patients indicated for cardiac resynchronization may also benefit from a novel therapy option the approval enables—left bundle branch optimized cardiac resynchronization therapy (LOT-CRT), which combines conduction system pacing with left-ventricular pacing to further improve patient outcomes.
“Conduction system pacing is a rapidly growing therapy for patients who need a pacemaker. Now, patients who require a defibrillator and pacing have an option that can safely deliver life-saving defibrillation therapy and activate the heart’s natural electrical system to enable a more synchronous, physiologic pattern,” says Trevor Cook, vice president and general manager of the Defibrillation Solutions business at Medtronic, in a release. “This approval underscores the versatility of the OmniaSecure defibrillation lead and supports its use across a variety of implant approaches to best serve a broad range of patients.”
Built on a Proven Lumenless Design
The OmniaSecure lead is engineered based on the Medtronic SelectSecure Model 3830 pacing lead, which became the first pacing lead to receive FDA approval for conduction system pacing in 2022 and has since been implanted in more than one million patients globally. Like its predecessor, the OmniaSecure lead features lumenless construction—meaning no hollow channel runs through the interior—which allows catheter-based delivery for precise placement in either the right ventricle or the LBB area, according to the release.
Traditional pacing methods are sometimes associated with cardiomyopathy or other complications. Conduction system pacing works by tapping into the heart’s existing electrical conduction pathway, offering a more physiologic activation pattern intended to reduce those risks, according to the release.
The OmniaSecure lead is compatible with Medtronic’s Cobalt and Crome family of transvenous ICD and CRT-D devices. For patients who do not require chronic pacing, the Medtronic extravascular Aurora EV-ICD system offers an alternative—delivering protection as the only ICD with a defibrillation lead positioned outside the vascular space that also provides anti-tachycardia pacing (ATP) in a single device, according to Medtronic.
LEADR LBBAP Trial Data Supported the Approval
FDA approval for the expanded LBB area indication was supported by data from the global LEADR LBBAP trial (Lead Evaluation for Defibrillation and Reliability in Left Bundle Branch Area Pacing). Late-breaking results presented at Heart Rhythm 2025 and published in Heart Rhythm showed a 100% defibrillation success rate at implant and a low OmniaSecure-related major complication rate of 2.1% at three months when the lead was placed in the LBB area, according to the release. Longer-term LBB area and LOT-CRT results from the study are expected to be presented at HRS 2026.
“The robust body of clinical evidence from the LEADR LBBAP study supports the OmniaSecure defibrillation lead as a safe and effective way to activate physiologic pacing through the left bundle branch area or in applications like LOT-CRT, while also demonstrating reliable defibrillation success,” says Pugazhendhi Vijayaraman, MD, cardiac electrophysiologist at Geisinger Wyoming Valley Medical Center in Wilkes-Barre, Pa, and principal investigator of the LEADR LBBAP study, in a release.
Expanding the Conduction System Pacing Portfolio
The approval adds to a growing portfolio of FDA-cleared lumenless leads and accessories that support conduction system pacing. Alongside the OmniaSecure lead, the portfolio includes the SelectSecure Model 3830 pacing lead, the recently cleared C320LBB delivery catheter, and the 5944RL rotatable connector.
The OmniaSecure defibrillation lead had already launched commercially in the US in January 2026 following initial FDA approval for placement in traditional right ventricular locations. The LBB area approval represents an expanded indication for a product already in clinical use.