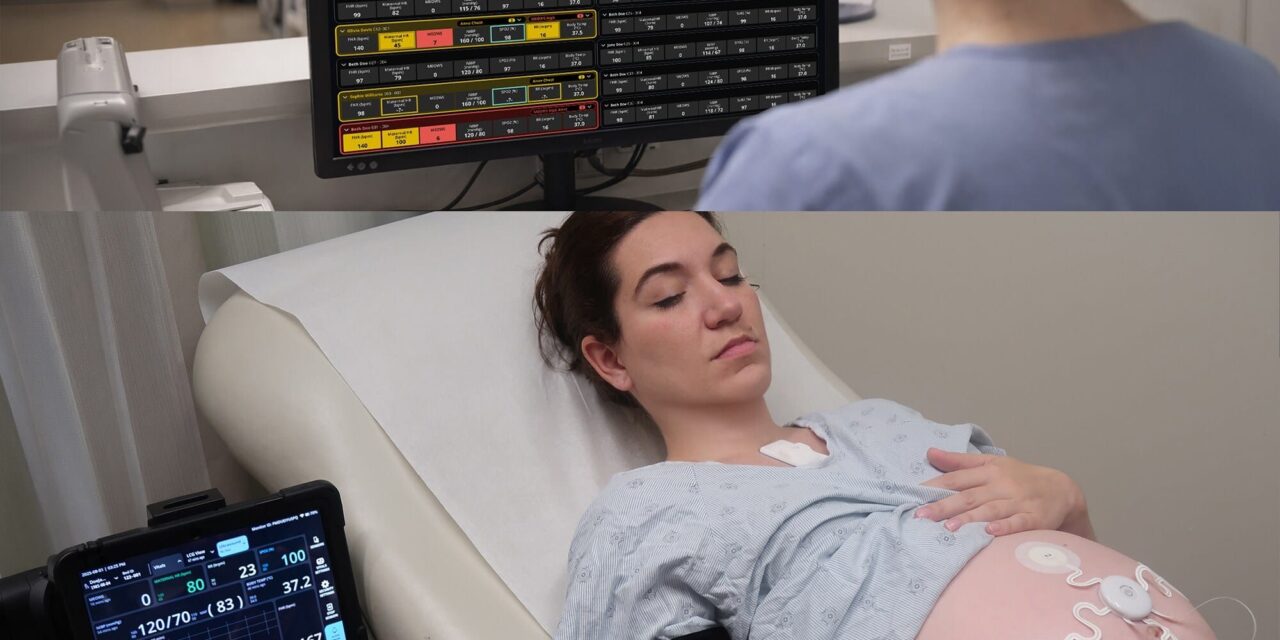
The wearable sensor system provides continuous, simultaneous monitoring of maternal and fetal vital signs to improve clinical evaluation.
The Food and Drug Administration (FDA) has granted 510(k) clearance to Sibel Health’s ANNE Maternal, a wireless maternal-fetal monitoring platform designed for continuous and simultaneous monitoring of maternal and fetal health.
The platform uses flexible epidermal wearable sensors to stream maternal vital signs, such as heart rate, respiratory rate, blood oxygen saturation, and skin temperature. The system also tracks fetal heart rate and uterine contractions without the use of wired equipment.
The platform includes an integrated electronic Modified Early Obstetric Warning System that provides automated signals to clinicians when parameters require prompt evaluation. It is also compatible with wireless point-of-care ultrasound.
“What makes this platform truly unique is its dual mandate: It meets the rigorous clinical standards demanded by the FDA while also being designed for deployment in the world’s most resource-limited settings,” says Steve Xu, MD, ceo and co-founder of Sibel Health, in a release.
The clearance arrives as the US maternal mortality rate stands at 22 deaths per 100,000 live births, which is the highest among high-income nations, according to a release from Sibel Health. For four years, the March of Dimes has issued the US a D+ grade for maternal and infant health.
The development of the platform was supported by a $17.5 million grant from the Gates Foundation. The technology is currently deployed in research partnerships in India, Pakistan, Nigeria, and Rwanda. Sibel Health also received a $5 million grant to develop future artificial intelligence features for the system.
“In our setting, the clinician-to-patient ratios are heavily constrained, and early detection of obstetric complications could be life-saving at scale,” says Kavita Bhatti, MD, professor and department chair of obstetrics and gynecology of Christian Medical College and Hospital Ludhiana, in a release.
“FDA clearance of ANNE Maternal reflects the kind of rigorous, science-driven review process that gives clinicians and patients confidence in the devices they rely on whether deployed in the US or in low resource settings,” says Indira Malladi, senior regulatory affairs specialist and lead regulatory author of the 510(k) submission, in a release.
Photo caption: ANNE Maternal
Photo credit: Sibel Health