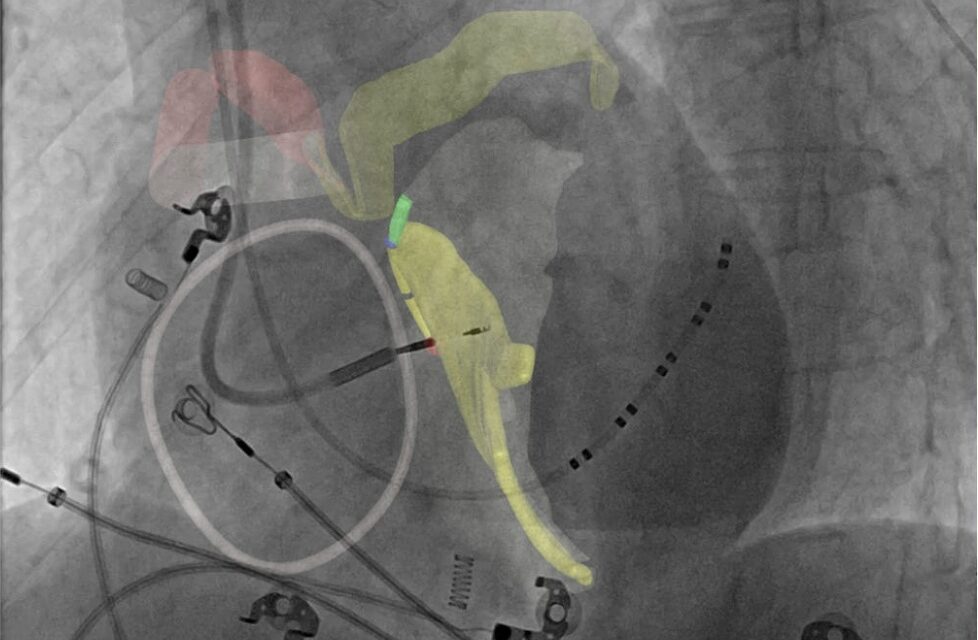
The CTA-based system creates a patient-specific 3D map of the cardiac conduction system to support planning and guidance during procedures such as TAVR and conduction system pacing.
Cara Medical Ltd announced that the US Food and Drug Administration has granted 510(k) clearance for the CARA System, a computed tomography angiography (CTA)-based platform designed to provide noninvasive, patient-specific three-dimensional visualization of the cardiac conduction system.
The CARA System, which received FDA Breakthrough Device Designation last year, enables visualization of the cardiac conduction system relative to surrounding heart structures during structural heart and pacing procedures.
The system is intended to support the planning and guidance of medical interventions in areas known to contain or lie near the cardiac conduction system. These include procedures such as transcatheter aortic valve replacement (TAVR) as well as interventions in which physicians aim to deliver therapy to the conduction system or a targeted location within it, including conduction system pacing.
The platform includes two components:
- CARA Metis Simulator – a preprocedural planning software that uses CTA-based anatomical landmarks to identify the cardiac conduction axis and generate a personalized 3D map of the patient’s cardiac conduction system.
- CARA Atlas Navigator – an intraprocedural guidance platform that overlays the patient-specific conduction system model generated by Metis onto live fluoroscopic images to assist physicians during image-guided interventions.
“This FDA clearance represents a significant milestone for Cara Medical,” said Shlomo Ben-Haim, MD, founder and chairman of Cara Medical Ltd, in a release. “We look forward to launching the product in the United States and providing physicians with a validated tool designed to enhance visualization of the cardiac conduction axis. Our growing body of clinical publications highlights Cara’s potential role in supporting procedural precision during TAVR and other valve interventions, and in facilitating physiologic lead targeting during conduction system pacing.”
According to the company, the CARA System runs on a standard workstation, integrates with existing fluoroscopy systems, and does not have direct patient contact. The system uses image processing and AI/ML-based algorithms to support CT segmentation, fluoroscopic metadata extraction, and catheter detection, with outputs subject to physician review and confirmation.
Cara Medical says it expects to begin its US commercial launch in the coming months.
Photo caption: CARA Atlas intra-procedural fluoroscopic overlay displaying the patient-specific cardiac conduction map generated by CARA Metis.
Photo credit: Cara Medical