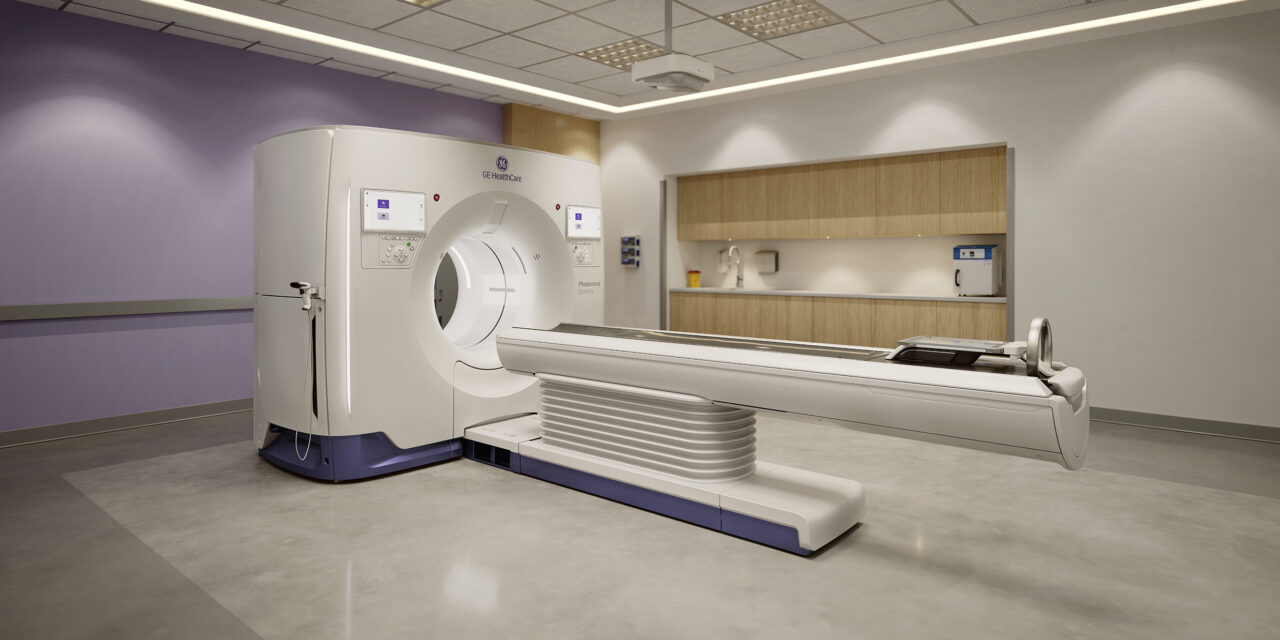
The cleared system introduces photon-counting CT designed to capture spectral data in a single scan and reduce protocol complexity.
GE HealthCare has received 510(k) clearance from the US Food and Drug Administration (FDA) for its Photonova Spectra photon-counting computed tomography system. The clearance follows the system’s debut at the Radiological Society of North America (RSNA) 2025 Annual Meeting.
Photonova Spectra is a spectral photon-counting CT system with 8-bin energy resolution, using the company’s Deep Silicon detector technology. It is designed as a single-scan workflow intended to reduce the need for multiple protocols across CT exams.
The system is part of GE HealthCare’s broader research and development efforts, which the company has said include a multibillion-dollar investment aimed at expanding its imaging portfolio.
Photon-counting CT differs from conventional CT systems in that it directly counts individual X-ray photons and measures their energy, rather than converting them into visible light. This approach can provide higher spectral and spatial resolution and support improved tissue characterization.
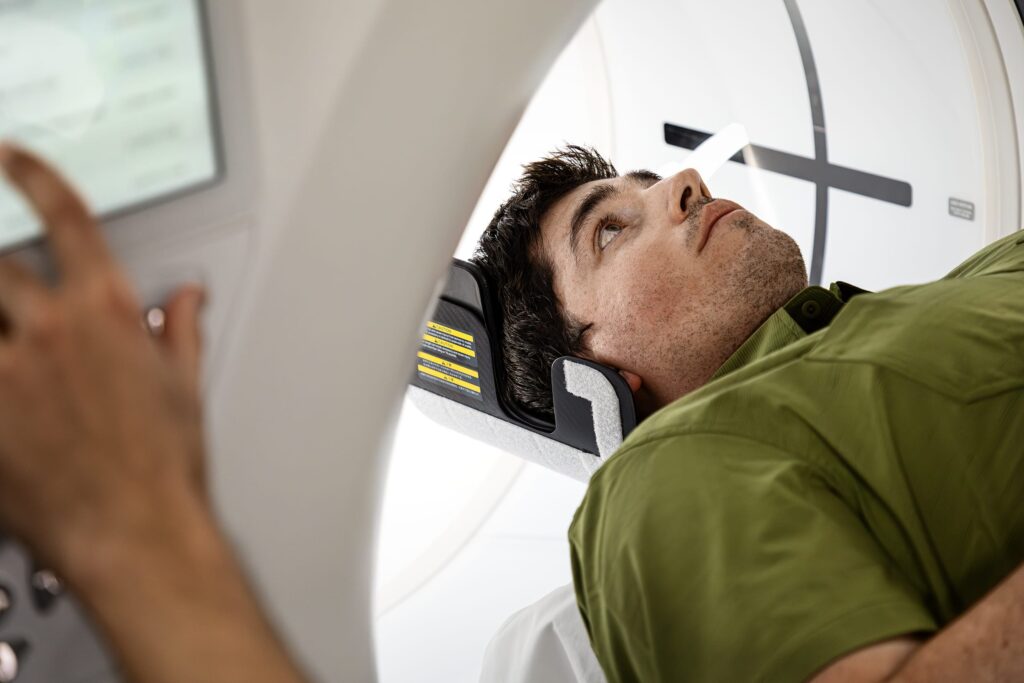
“As clinicians across the United States face rising volumes and increasing diagnostic complexity, technology must do more than capture images; it must simplify decision-making and strengthen performance across the enterprise,” says Catherine Estrampes, president and CEO of US and Canada at GE HealthCare, in a release. “Photonova Spectra is designed to deliver rich clinical insights in every scan and help alleviate cognitive burden for care teams.”
According to GE HealthCare, the Deep Silicon detector is designed to enable more precise measurement of photon energy, which may support advanced image reconstruction and material differentiation. The system can distinguish between materials such as iodine, calcium, and fat and is intended for use across multiple clinical areas, including oncology, cardiology, neurology, and musculoskeletal imaging.
The system also incorporates GPU-based computing technology to process the larger data volumes associated with photon-counting CT. GE HealthCare states that this architecture is designed to support image reconstruction and workflow efficiency.
Workflow features include a one-scan approach that captures spectral and spatial data simultaneously, along with automated positioning and reconstruction tools intended to support consistency across exams.
GE HealthCare says the system is being evaluated at institutions including the University of Wisconsin–Madison and Stanford Medicine, where research is focused on image quality, workflow optimization, and potential clinical applications.
The company indicates that Photonova Spectra is designed to be installed in existing CT environments with minimal modifications. Following FDA clearance, GE HealthCare says it will begin preparing for commercial availability in the United States.
Photo caption: Photonova Spectra
Photo credit: GE HealthCare